AUCTORES
Globalize your Research
Research Article
*Corresponding Author: Ampati Srinivas, Deportment of Medicinal chemistry, Unity college of Pharmacy, Bongir, India.
Citation: Ampati Srinivas Kokkula pavan Kumar and Prasad Garrepally, Subcutaneous DL Technique Has Proven To Be an Adequate Host for Human Embryonic Stem Cells, DOI: 10.31579/2643-1912/004
Copyright: © 2019 Ampati Srinivas, This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 18 March 2019 | Accepted: 09 April 2019 | Published: 12 April 2019
Keywords: islet Transplantation; embryonic stem cells; cell engraftment; cell maturation
Islet transplantation has become an important treatment modality for Type 1 Diabetes Mellitus (T1DM); nonetheless, the procedure may be limited by donor availability. An alternative has been the increasing use of cellular therapies derived from human Embryonic Stem Cells (hESC), showing very promising results in maturation, yield and ultimately, in insulin secretion in response to adequate stimuli. We recently developed a new technique for cellular transplantation under the skin. This manuscript evaluates the capabilities of the pre-vascularized Device-Less (DL) site to allow transplantation of Pancreatic Endoderm (PE) cells differentiated from hESC to treat diabetes mellitus. Fifty immunodeficient mice, n = 25 diabetic and n = 25 non-diabetic, were transplanted with PE cells. Animals were followed for 22 weeks and grafts were retrieved to evaluate engraftment and subsequent maturation. Diabetic mice showed slightly better engraftment (48% vs. 36%, p = 0.19) and secreted higher concentration of human C-peptide upon glucose stimulation (0.32 ± 0.15 ng/mL vs. 0.13 ± 0.09 ng/mL, p = 0.30), although differences were not significant. This maturation was not sufficient to successfully reverse diabetes. Monomorphic cystic changes were detected in 12% and 8%, respectively (diabetics vs. non-diabetics, p = 0.32) and all grafts seemed to be adequately contained by the surrounding collagen wall within the DL space. Our findings support the capabilities of the DL site to host PE cells and allow safe maturation as a new strategy to treat diabetes.
The recent advances in immunotherapy have allowed Islet Transplantation (IT) to become a mainstay treatment for Type 1 Diabetes Mellitus (T1DM). Today, the procedure is safer and long-term graft survival is comparable to that of pancreas transplant alone, with a reduced risk for complications [1,2]. Nonetheless, the IT procedure is limited by donor availability and usage. Significant variability is associated with this treatment modality and many factors may affect the successful utilization of a donated pancreas. In fact, the entire donation-transplant process depends upon many variables related to the donor clinical characteristics, the type of donation (living, brain death, cardiac death, etc.), the outcomes of islet isolation, and recipient characteristics. As a consequence, the process is not always efficient and like other transplant types, the demand may surpass the available donation pool.
An alternative to IT may be to use renewable sources for insulin secretion from proliferative stem cell populations. In particular, research using insulin-producing cells derived from human embryonic stem cells (hESC) has shown very promising results in maturation yield and ultimately, in insulin secretion in response to adequate stimuli [3-6]. The focus is now on optimizing the existing differentiation protocols to allow for a successful and stable diabetes reversal. However, finding the most efficient transplant site remains a dilemma given the infusion volume needed at the time of transplant and the potential need for graft retrieval in the event of tumor formation [7,8]. These reasons are a deterrent to use the conventional intra portal route for this transplantation modality.
Our group recently described a novel pre-vascularized Device-Less (DL) technique for cell transplantation in the subcutaneous space [9]. This approach was successful in reversing diabetes with mouse and human islets and is currently being used for other cell therapies. We herein describe the use of the DL technique to safely allow engraftment and maturation of Pancreatic Endoderm (PE) cells derived from a hESC line in an experimental xeno-transplant model of diabetes.
Human Embryonic Stem Cells-derived Pancreatic Endoderm
Pancreatic Endoderm (PE) cells derived from a human embryonic cell line were kindly provided by Drs. M.C. Nostro and G. Keller at the McEwen Centre for Regenerative Medicine in Toronto. Their differentiation protocol uses a combination of cytokines and small molecules to simulate pancreatic development and produces multipotent pancreatic progenitor cells with the potential to differentiate into all pancreatic lineages [10,11]. At the time of transplant, cells were harvested and shipped overnight to Edmonton for immediate implantation.
Transplantation of PE cells
Immunodeficient 8-12 week B6.129S7-Rag1tm1Mom mice (Jackson Laboratory, Bar Harbor, ME, USA) were used for all experiments. Animals (n = 50) were housed under conventional conditions with access to food and water ad libitum and their care was in accordance with guidelines approved by the Canadian Council on Animal Care.
The DL space was created as previously reported by inserting a nylon catheter subcutaneously in the left lower abdomen and left for five weeks before transplant [9].Diabetes was chemically induced by intraperitoneally injecting 180 mg/kg of streptozotocin (STZ; Sigma-Aldrich, ON, Canada) in half of the recipients, one week prior to transplantation. Mice were considered diabetic after two consecutive blood glucose measurements ≥ 11.3 mmol/L (350 mg/dL).
Two groups of mice (diabetics and non-diabetics, n = 25/group) were transplanted with approximately 7×106 PE cells using the DL technique. Animals in the diabetic group also received two consecutive insulin-releasing pellets (LinBit®; LinShin Canada Inc. Toronto, ON, Canada - ~0.1 U insulin/24 hours/30 days) to maintain health for the duration of the study (160 days). A separate group of four mice (two diabetics and two non-diabetics) were transplanted with same amount of PE cells and sacrificed four week post-transplant for early assessment of the graft. All mice were continuously monitored for general health, weight gain and non-fasting blood glucose, as well as the occurrence of tumor formation.
C-peptide Measurements
Blood samples were also obtained at post-transplant week 4, 8, 12, 16, 20 and 22 to quantify stimulated human C-peptide concentration in plasma. Mice from both groups were fasted overnight and whole blood was collected after intraperitoneal injection of glucose (2 g/kg). Quantification of C-peptide was performed using human-specific ultrasensitive ELISA (Mercodia, Uppsala, Sweden. Detection range: 5 - 280 pmol/L (0.015 - 0.85 ng/mL).
Histology
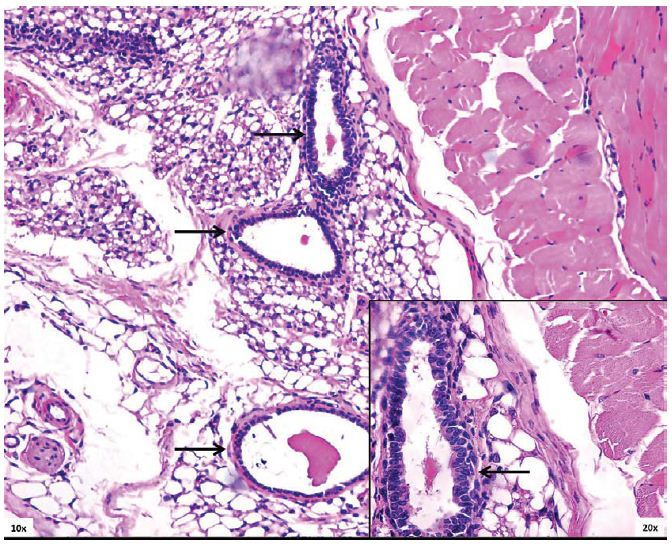
Engrafted cells were analyzed at early (four weeks post-transplant) and at the end of the study. Hematoxylin and eosin (H&E) and Masson’s trichrome stains were used to visualize the grafts on abdominal wall sections and to assess tumor boundaries. Immunofluorescence was used to evaluate endocrine secretory function of long-term engrafted cells using anti-insulin (Dako A0082 – Alexa 568) and anti-glucagon (Abcam – Vector Fl-1000) antibodies. The procedure followed previously established methodology [9] and it included deparaffinization, primary and secondary antibody treatment and counter stain with DAPI (Invitrogen Molecular Probes. Eugene, Oregon). Slides were visualized using a fluorescent microscope with appropriate filters and AxioVision imaging software (Carl Zeiss Microscopy GmbH. Jena, Germany).
Transplantation of Human Islets
In parallel, human islets were transplanted into 8-12 week B6.129S7-Rag1tm1Mom diabetic mice and monitored for 22 weeks to compare human C-peptide secretion levels to those achieved by the study PE cells. The Clinical Islet Transplantation laboratory at the University of Alberta kindly provided human islets after the process of donation, isolation and culture, as reported in previous publications [12]. Permission for these studies was granted by the Health Research Ethics Board of the University of Alberta, Edmonton, Alberta, Canada, and after written permission was obtained from donor families.
Mice were rendered diabetic by intraperitoneal injection of 180 mg/kg streptozotocin (STZ, Sigma-Aldrich, ON, Canada). Animals were considered diabetic after two consecutive blood glucose measurements ≥ 11.3 mmol/L (350 mg/dL). Recipients (n = 6 per group) received 0 IEQ (Sham and STZ), 1,000 IEQ and 3,000 IEQ human islets from three different isolations. Islets from each isolation were randomly allocated to each group and transplanted under the kidney capsule as previously described [13]. Mice in the Sham group were not diabetic and underwent the transplant procedure, but only received a saline solution under the kidney capsule. Animals in the STZ group were chemically-induced diabetics and did not receive transplant, remaining diabetic throughout the entire study.
Animals were periodically monitored for general health, weight and blood glucose until endpoint (22 weeks) when blood samples were taken to determine basal and stimulated human C-peptide levels.
A total of 54 mice received 7×106 PE cells/mouse subcutaneously, using the DL technique. Four weeks after transplantation, a viable graft was found in all four sacrificed animals, showing clear features of ductal formations (Figure 1). However, all animals remained diabetic and no detectable human C-peptide was found at this early time point (data not shown).
Twenty weeks after transplantation stimulated C-peptide was detected in both groups and continued to increase until the end of the study. Positive stimulated C-peptide was found in 12 of 25 (48%) mice in the diabetic group vs. 9 of 25 (36%), in non-diabetic (p = 0.19). Mean stimulated C-peptide concentrations at 22 weeks were higher in the diabetic group although differences were not statistically significant (0.32 ± 0.15 ng/mL vs. 0.13 ± 0.09 ng/mL, p = 0.30) (Figure 2A).
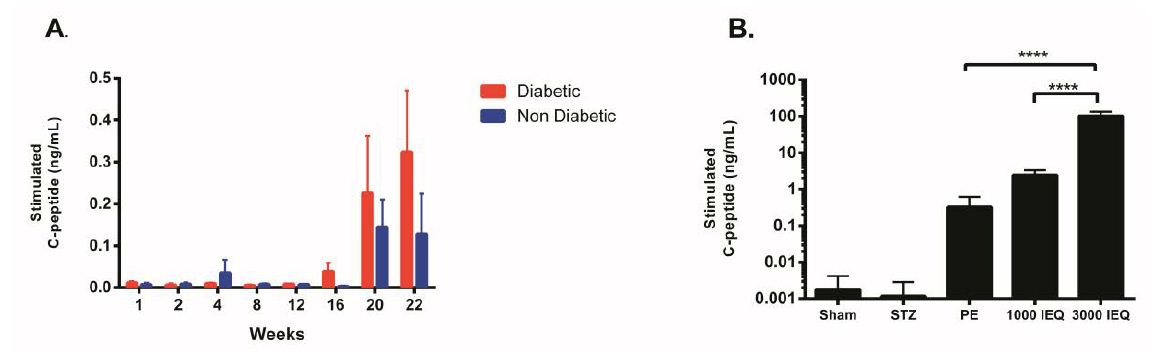
Figure 2: Stimulated C-peptide secretion from transplanted pancreatic endoderm (PE) cells; A. Secretory profile over time for diabetic and nondiabetic mice, showing detectable C-peptide beyond 20 weeks post-transplant; B. Comparison of C-peptide secretory capabilities of transplanted PE cells vs. human islets at minimal and full dose transplanted in immunodeficient mice. A Sham and STZ (diabetic, non-transplanted) groups are included for comparison and specificity of the assay.
Our findings corroborate the utility of the DL technique to facilitate cell therapies. In this case, the aim was to engraft and mature PE cells derived from hESC, and measure indicators towards diabetes reversal.
In our study design we evaluated the potential effect of underlying hyperglycemia for engraftment and maturation of PE cells based on published evidence for accelerated maturation under a chronic hyperglycemic environment [14]. Results confirmed an increased trend in both, maturation and mean concentration of stimulated human C-peptide levels measured at 22 weeks in diabetic mice. Differences however, did not reach statistical significance. C-peptide positive measurements were specific to engrafted cells from human origin, proving to be a valuable tool for assessment of maturation of transplanted PEs.
The process of effective differentiation of hESC is very complex and yet to be fully elucidated. Many authors agree on the multiple hurdles these cells encounter in the process of maturation and only recently, successful in vivo maturation have been reported with adequate glucose-response and occasionally, diabetes reversal [15-19].
Consistent with previous studies, the PE cells we tested in our experiments require a long (more than five months) in vivo maturation period [10,17,20,21].
Despite observing adequate engraftment in almost half of the animals, glucagon staining was predominant in most of the histology samples and positive insulin cells were only occasionally found, which is consistent with the low levels of stimulated c-peptide detected at week 22, as well as failure to correct hyperglycemia. We speculate that longer in vivo maturation or a higher number of hESC-derived PE cells at the time of transplantation may be required to normalize glucose control in diabetic mice, although a more prominent insulin staining has been previously reported when transplanting these cells in the kidney subcapsular space and mammary fat pad [10]. Further experimentation will definitely be required to fully understand the maturation process in this new transplant site.
One of the main limitations for the use of hESC is the inherit risk for teratoma transformation [7]. This is one of the rationales for using alternative transplant sites like the DL technique where a dysfunctional/transforming graft may be easily retrieved. Current differentiation protocols are now focused on producing hESC preparations with a high grade of purity to avoid residual undifferentiated cells, which could potentially lead to tumor formation [22]. In our series of transplants benign monomorphic cystic formations were present in 8 - 12% of cases and no teratoma was detected.
An interesting finding was that resulting cysts were successfully contained by the peripheral collagen wall present in the DL space during the 22-week observation period. This resulted in a restrictive effect similar to that present in other physical devices [14,18]. However, the real restraining capabilities in the settings of a true teratoma formation are still to be proven.
In conclusion, our subcutaneous DL technique has proven to be an adequate host for these human embryonic stem cells - derived pancreatic endoderm, allowing effective engraftment, maturation and added protection against tumor formations. This is certainly an important field of application for this technique and a starting point for further experimentation with improved cell preparation and transplant protocols.
Clearly Auctoresonline and particularly Psychology and Mental Health Care Journal is dedicated to improving health care services for individuals and populations. The editorial boards' ability to efficiently recognize and share the global importance of health literacy with a variety of stakeholders. Auctoresonline publishing platform can be used to facilitate of optimal client-based services and should be added to health care professionals' repertoire of evidence-based health care resources.

Journal of Clinical Cardiology and Cardiovascular Intervention The submission and review process was adequate. However I think that the publication total value should have been enlightened in early fases. Thank you for all.

Journal of Women Health Care and Issues By the present mail, I want to say thank to you and tour colleagues for facilitating my published article. Specially thank you for the peer review process, support from the editorial office. I appreciate positively the quality of your journal.
Journal of Clinical Research and Reports I would be very delighted to submit my testimonial regarding the reviewer board and the editorial office. The reviewer board were accurate and helpful regarding any modifications for my manuscript. And the editorial office were very helpful and supportive in contacting and monitoring with any update and offering help. It was my pleasure to contribute with your promising Journal and I am looking forward for more collaboration.

We would like to thank the Journal of Thoracic Disease and Cardiothoracic Surgery because of the services they provided us for our articles. The peer-review process was done in a very excellent time manner, and the opinions of the reviewers helped us to improve our manuscript further. The editorial office had an outstanding correspondence with us and guided us in many ways. During a hard time of the pandemic that is affecting every one of us tremendously, the editorial office helped us make everything easier for publishing scientific work. Hope for a more scientific relationship with your Journal.

The peer-review process which consisted high quality queries on the paper. I did answer six reviewers’ questions and comments before the paper was accepted. The support from the editorial office is excellent.

Journal of Neuroscience and Neurological Surgery. I had the experience of publishing a research article recently. The whole process was simple from submission to publication. The reviewers made specific and valuable recommendations and corrections that improved the quality of my publication. I strongly recommend this Journal.

Dr. Katarzyna Byczkowska My testimonial covering: "The peer review process is quick and effective. The support from the editorial office is very professional and friendly. Quality of the Clinical Cardiology and Cardiovascular Interventions is scientific and publishes ground-breaking research on cardiology that is useful for other professionals in the field.

Thank you most sincerely, with regard to the support you have given in relation to the reviewing process and the processing of my article entitled "Large Cell Neuroendocrine Carcinoma of The Prostate Gland: A Review and Update" for publication in your esteemed Journal, Journal of Cancer Research and Cellular Therapeutics". The editorial team has been very supportive.

Testimony of Journal of Clinical Otorhinolaryngology: work with your Reviews has been a educational and constructive experience. The editorial office were very helpful and supportive. It was a pleasure to contribute to your Journal.

Dr. Bernard Terkimbi Utoo, I am happy to publish my scientific work in Journal of Women Health Care and Issues (JWHCI). The manuscript submission was seamless and peer review process was top notch. I was amazed that 4 reviewers worked on the manuscript which made it a highly technical, standard and excellent quality paper. I appreciate the format and consideration for the APC as well as the speed of publication. It is my pleasure to continue with this scientific relationship with the esteem JWHCI.

This is an acknowledgment for peer reviewers, editorial board of Journal of Clinical Research and Reports. They show a lot of consideration for us as publishers for our research article “Evaluation of the different factors associated with side effects of COVID-19 vaccination on medical students, Mutah university, Al-Karak, Jordan”, in a very professional and easy way. This journal is one of outstanding medical journal.
Dear Hao Jiang, to Journal of Nutrition and Food Processing We greatly appreciate the efficient, professional and rapid processing of our paper by your team. If there is anything else we should do, please do not hesitate to let us know. On behalf of my co-authors, we would like to express our great appreciation to editor and reviewers.

As an author who has recently published in the journal "Brain and Neurological Disorders". I am delighted to provide a testimonial on the peer review process, editorial office support, and the overall quality of the journal. The peer review process at Brain and Neurological Disorders is rigorous and meticulous, ensuring that only high-quality, evidence-based research is published. The reviewers are experts in their fields, and their comments and suggestions were constructive and helped improve the quality of my manuscript. The review process was timely and efficient, with clear communication from the editorial office at each stage. The support from the editorial office was exceptional throughout the entire process. The editorial staff was responsive, professional, and always willing to help. They provided valuable guidance on formatting, structure, and ethical considerations, making the submission process seamless. Moreover, they kept me informed about the status of my manuscript and provided timely updates, which made the process less stressful. The journal Brain and Neurological Disorders is of the highest quality, with a strong focus on publishing cutting-edge research in the field of neurology. The articles published in this journal are well-researched, rigorously peer-reviewed, and written by experts in the field. The journal maintains high standards, ensuring that readers are provided with the most up-to-date and reliable information on brain and neurological disorders. In conclusion, I had a wonderful experience publishing in Brain and Neurological Disorders. The peer review process was thorough, the editorial office provided exceptional support, and the journal's quality is second to none. I would highly recommend this journal to any researcher working in the field of neurology and brain disorders.

Dear Agrippa Hilda, Journal of Neuroscience and Neurological Surgery, Editorial Coordinator, I trust this message finds you well. I want to extend my appreciation for considering my article for publication in your esteemed journal. I am pleased to provide a testimonial regarding the peer review process and the support received from your editorial office. The peer review process for my paper was carried out in a highly professional and thorough manner. The feedback and comments provided by the authors were constructive and very useful in improving the quality of the manuscript. This rigorous assessment process undoubtedly contributes to the high standards maintained by your journal.

International Journal of Clinical Case Reports and Reviews. I strongly recommend to consider submitting your work to this high-quality journal. The support and availability of the Editorial staff is outstanding and the review process was both efficient and rigorous.

Thank you very much for publishing my Research Article titled “Comparing Treatment Outcome Of Allergic Rhinitis Patients After Using Fluticasone Nasal Spray And Nasal Douching" in the Journal of Clinical Otorhinolaryngology. As Medical Professionals we are immensely benefited from study of various informative Articles and Papers published in this high quality Journal. I look forward to enriching my knowledge by regular study of the Journal and contribute my future work in the field of ENT through the Journal for use by the medical fraternity. The support from the Editorial office was excellent and very prompt. I also welcome the comments received from the readers of my Research Article.

Dear Erica Kelsey, Editorial Coordinator of Cancer Research and Cellular Therapeutics Our team is very satisfied with the processing of our paper by your journal. That was fast, efficient, rigorous, but without unnecessary complications. We appreciated the very short time between the submission of the paper and its publication on line on your site.

I am very glad to say that the peer review process is very successful and fast and support from the Editorial Office. Therefore, I would like to continue our scientific relationship for a long time. And I especially thank you for your kindly attention towards my article. Have a good day!

"We recently published an article entitled “Influence of beta-Cyclodextrins upon the Degradation of Carbofuran Derivatives under Alkaline Conditions" in the Journal of “Pesticides and Biofertilizers” to show that the cyclodextrins protect the carbamates increasing their half-life time in the presence of basic conditions This will be very helpful to understand carbofuran behaviour in the analytical, agro-environmental and food areas. We greatly appreciated the interaction with the editor and the editorial team; we were particularly well accompanied during the course of the revision process, since all various steps towards publication were short and without delay".

I would like to express my gratitude towards you process of article review and submission. I found this to be very fair and expedient. Your follow up has been excellent. I have many publications in national and international journal and your process has been one of the best so far. Keep up the great work.

We are grateful for this opportunity to provide a glowing recommendation to the Journal of Psychiatry and Psychotherapy. We found that the editorial team were very supportive, helpful, kept us abreast of timelines and over all very professional in nature. The peer review process was rigorous, efficient and constructive that really enhanced our article submission. The experience with this journal remains one of our best ever and we look forward to providing future submissions in the near future.

I am very pleased to serve as EBM of the journal, I hope many years of my experience in stem cells can help the journal from one way or another. As we know, stem cells hold great potential for regenerative medicine, which are mostly used to promote the repair response of diseased, dysfunctional or injured tissue using stem cells or their derivatives. I think Stem Cell Research and Therapeutics International is a great platform to publish and share the understanding towards the biology and translational or clinical application of stem cells.

I would like to give my testimony in the support I have got by the peer review process and to support the editorial office where they were of asset to support young author like me to be encouraged to publish their work in your respected journal and globalize and share knowledge across the globe. I really give my great gratitude to your journal and the peer review including the editorial office.

I am delighted to publish our manuscript entitled "A Perspective on Cocaine Induced Stroke - Its Mechanisms and Management" in the Journal of Neuroscience and Neurological Surgery. The peer review process, support from the editorial office, and quality of the journal are excellent. The manuscripts published are of high quality and of excellent scientific value. I recommend this journal very much to colleagues.

Dr.Tania Muñoz, My experience as researcher and author of a review article in The Journal Clinical Cardiology and Interventions has been very enriching and stimulating. The editorial team is excellent, performs its work with absolute responsibility and delivery. They are proactive, dynamic and receptive to all proposals. Supporting at all times the vast universe of authors who choose them as an option for publication. The team of review specialists, members of the editorial board, are brilliant professionals, with remarkable performance in medical research and scientific methodology. Together they form a frontline team that consolidates the JCCI as a magnificent option for the publication and review of high-level medical articles and broad collective interest. I am honored to be able to share my review article and open to receive all your comments.

“The peer review process of JPMHC is quick and effective. Authors are benefited by good and professional reviewers with huge experience in the field of psychology and mental health. The support from the editorial office is very professional. People to contact to are friendly and happy to help and assist any query authors might have. Quality of the Journal is scientific and publishes ground-breaking research on mental health that is useful for other professionals in the field”.

Dear editorial department: On behalf of our team, I hereby certify the reliability and superiority of the International Journal of Clinical Case Reports and Reviews in the peer review process, editorial support, and journal quality. Firstly, the peer review process of the International Journal of Clinical Case Reports and Reviews is rigorous, fair, transparent, fast, and of high quality. The editorial department invites experts from relevant fields as anonymous reviewers to review all submitted manuscripts. These experts have rich academic backgrounds and experience, and can accurately evaluate the academic quality, originality, and suitability of manuscripts. The editorial department is committed to ensuring the rigor of the peer review process, while also making every effort to ensure a fast review cycle to meet the needs of authors and the academic community. Secondly, the editorial team of the International Journal of Clinical Case Reports and Reviews is composed of a group of senior scholars and professionals with rich experience and professional knowledge in related fields. The editorial department is committed to assisting authors in improving their manuscripts, ensuring their academic accuracy, clarity, and completeness. Editors actively collaborate with authors, providing useful suggestions and feedback to promote the improvement and development of the manuscript. We believe that the support of the editorial department is one of the key factors in ensuring the quality of the journal. Finally, the International Journal of Clinical Case Reports and Reviews is renowned for its high- quality articles and strict academic standards. The editorial department is committed to publishing innovative and academically valuable research results to promote the development and progress of related fields. The International Journal of Clinical Case Reports and Reviews is reasonably priced and ensures excellent service and quality ratio, allowing authors to obtain high-level academic publishing opportunities in an affordable manner. I hereby solemnly declare that the International Journal of Clinical Case Reports and Reviews has a high level of credibility and superiority in terms of peer review process, editorial support, reasonable fees, and journal quality. Sincerely, Rui Tao.

Clinical Cardiology and Cardiovascular Interventions I testity the covering of the peer review process, support from the editorial office, and quality of the journal.

Clinical Cardiology and Cardiovascular Interventions, we deeply appreciate the interest shown in our work and its publication. It has been a true pleasure to collaborate with you. The peer review process, as well as the support provided by the editorial office, have been exceptional, and the quality of the journal is very high, which was a determining factor in our decision to publish with you.
The peer reviewers process is quick and effective, the supports from editorial office is excellent, the quality of journal is high. I would like to collabroate with Internatioanl journal of Clinical Case Reports and Reviews journal clinically in the future time.

Clinical Cardiology and Cardiovascular Interventions, I would like to express my sincerest gratitude for the trust placed in our team for the publication in your journal. It has been a true pleasure to collaborate with you on this project. I am pleased to inform you that both the peer review process and the attention from the editorial coordination have been excellent. Your team has worked with dedication and professionalism to ensure that your publication meets the highest standards of quality. We are confident that this collaboration will result in mutual success, and we are eager to see the fruits of this shared effort.

Dear Dr. Jessica Magne, Editorial Coordinator 0f Clinical Cardiology and Cardiovascular Interventions, I hope this message finds you well. I want to express my utmost gratitude for your excellent work and for the dedication and speed in the publication process of my article titled "Navigating Innovation: Qualitative Insights on Using Technology for Health Education in Acute Coronary Syndrome Patients." I am very satisfied with the peer review process, the support from the editorial office, and the quality of the journal. I hope we can maintain our scientific relationship in the long term.
Dear Monica Gissare, - Editorial Coordinator of Nutrition and Food Processing. ¨My testimony with you is truly professional, with a positive response regarding the follow-up of the article and its review, you took into account my qualities and the importance of the topic¨.

Dear Dr. Jessica Magne, Editorial Coordinator 0f Clinical Cardiology and Cardiovascular Interventions, The review process for the article “The Handling of Anti-aggregants and Anticoagulants in the Oncologic Heart Patient Submitted to Surgery” was extremely rigorous and detailed. From the initial submission to the final acceptance, the editorial team at the “Journal of Clinical Cardiology and Cardiovascular Interventions” demonstrated a high level of professionalism and dedication. The reviewers provided constructive and detailed feedback, which was essential for improving the quality of our work. Communication was always clear and efficient, ensuring that all our questions were promptly addressed. The quality of the “Journal of Clinical Cardiology and Cardiovascular Interventions” is undeniable. It is a peer-reviewed, open-access publication dedicated exclusively to disseminating high-quality research in the field of clinical cardiology and cardiovascular interventions. The journal's impact factor is currently under evaluation, and it is indexed in reputable databases, which further reinforces its credibility and relevance in the scientific field. I highly recommend this journal to researchers looking for a reputable platform to publish their studies.

Dear Editorial Coordinator of the Journal of Nutrition and Food Processing! "I would like to thank the Journal of Nutrition and Food Processing for including and publishing my article. The peer review process was very quick, movement and precise. The Editorial Board has done an extremely conscientious job with much help, valuable comments and advices. I find the journal very valuable from a professional point of view, thank you very much for allowing me to be part of it and I would like to participate in the future!”

Dealing with The Journal of Neurology and Neurological Surgery was very smooth and comprehensive. The office staff took time to address my needs and the response from editors and the office was prompt and fair. I certainly hope to publish with this journal again.Their professionalism is apparent and more than satisfactory. Susan Weiner

My Testimonial Covering as fellowing: Lin-Show Chin. The peer reviewers process is quick and effective, the supports from editorial office is excellent, the quality of journal is high. I would like to collabroate with Internatioanl journal of Clinical Case Reports and Reviews.

My experience publishing in Psychology and Mental Health Care was exceptional. The peer review process was rigorous and constructive, with reviewers providing valuable insights that helped enhance the quality of our work. The editorial team was highly supportive and responsive, making the submission process smooth and efficient. The journal's commitment to high standards and academic rigor makes it a respected platform for quality research. I am grateful for the opportunity to publish in such a reputable journal.
My experience publishing in International Journal of Clinical Case Reports and Reviews was exceptional. I Come forth to Provide a Testimonial Covering the Peer Review Process and the editorial office for the Professional and Impartial Evaluation of the Manuscript.
