AUCTORES
Globalize your Research
Research Article | DOI: https://doi.org/10.31579/2690-4861/604
1Univ. Lille, Inserm, CHU Lille U1189 - ONCO-THAI - Assisted Laser Therapy and Immunotherapy for Oncology, Lille, France.
2Univ. Caen Normandie, INSERM U1086 ANTICIPE (Interdisciplinary Research Unit for Cancers Prevention and Treatment), BioTICLA Laboratory (Precision Medicine for Ovarian Cancers), Caen, France.
3UNICANCER, Comprehensive Cancer Center François Baclesse, Caen, France.
4Department of Medical Oncology, Centre Henri Becquerel, Rouen, France.
5Univ. Lille, CNRS, Inserm, CHU Lille, UMR9020-U1277 - CANTHER – Cancer Heterogeneity Plasticity and Resistance to Therapies, F-59000 Lille, France.
6Université de Caen Normandie, Services Unit PLATON, ORGAPRED Core Facility, Caen, France.
*Corresponding Author: Camille Trioen, Univ. Lille, Inserm, CHU Lille U1189 - ONCO-THAI - Assisted Laser Therapy and Immunotherapy for Oncology, Lille, France.
Citation: Léa Boidin, Lucie Thorel, Camille Trioen, Olivier Morales, Nadira Delhem, et al, (2024), High Efficiency of a Folate Receptor Alpha Targeted Photodynamic Therapy on Epithelial Ovarian Cancer Patient-Derived Tumor Organoids Resistant to Parpi, International Journal of Clinical Case Reports and Reviews, 21(1); DOI:10.31579/2690-4861/604
Copyright: © 2024, Camille Trioen. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 08 November 2024 | Accepted: 04 December 2024 | Published: 31 December 2024
Keywords: photodynamic therapy; PARP inhibitors; patient-derived tumor organoids; ovarian cancer; folate receptor alpha
Epithelial Ovarian Cancer (EOC) is the seventh cause of cancer-related deaths in women. The inhibition of poly- ADP-ribose polymerase (PARP) by PARP inhibitors (PARPi), such as Olaparib, has considerably improved the outcome of patients with homologous recombination deficiency (HRD) status. However, the major toxicity of PARPi combined with the emergence of resistance has led to the development of innovative strategies that can be used in combination or alone in order to optimize treatment and improve patient prognosis. The aim of this study was to use EOC cell line and Patient-Derived Tumor Organoids (PDTO) to provide key information on the potential of an innovative treatment based on folate receptor alpha (FRα)-targeted PDT combined or not to PARPi, in the context of intrinsic or acquired drug resistance. Interestingly, we showed for the first time that organoids derived from EOC cell line and Patient-Derived Tumor Organoids (PDTO) resistant to PARPi exhibited an excellent sensitivity to Frα-targeted PDT. Furthermore, PDT alone is sufficient to induce PDTOs death and prior treatment with PARPi does not improve therapeutic efficacy. These results provide new perspectives about the potential of PDT to overcome resistance to conventional treatment which open new therapeutic option for EOC.
Ovarian carcinoma (OC) is a highly frequent condition associated with a poor prognosis, with around 200,000 deaths worldwide by 2022 [1]. Among OC, Epithelial Ovarian Cancer (EOC) is the most frequent subtype and the deadliest, representing 90% of OC cases and the most common EOC histological subtypes (70%) is the high grade serous ovarian carcinoma (HGSOC) [2]. Due to the lack of symptoms of EOC in early stages, 80% of patients are diagnosed with an advanced disease (stage III or IV according to the FIGO classification) [3]. In these advanced stages, widespread intra-abdominal disease with peritoneal metastases is often present and is characterized as peritoneal carcinosis [4].
The advent of poly-ADP-ribose polymerase inhibitors (PARPi) (e.g., Olaparib, Niraparib, Rucaparib) has dramatically changed the prognosis of patient with peritoneal carcinosis from epithelial origin. Around 50% of HGSOC present a homologous recombination (HR) deficiency (HRD) status, including 15%-20% of germline BRCA1/2 mutations [5]. PARP inhibition prevents the repair of DNA single-strand breaks (SSBs) by transforming them into double-strand breaks (DSBs) that cannot be repaired in an HRD context, thus leading to synthetic lethality. Somatic BRCA1/2 mutations, hypermethylation of the BRCA1 promoter and deficiency in other proteins involved in the HR pathways contribute to the remainder of HRD. Although these PARPi have provided clinical benefit among patients with germline and/or tumor BRCA mutation and/or HRD tumors [6], they are not devoid of toxicities and poorly tolerated. Furthermore, although some patients exhibit intrinsic resistance to PARPi, various acquired resistance mechanisms (e.g., BRCA reversion mutation, restoration of HR repair function, replication fork stabilization, epigenetic changes) can lead to disease progression during PARPi therapies [5,7–9]. Therefore, combinatorial therapeutics strategies are a cornerstone in cancer therapeutics that may be leveraged to (i) enable dose reductions of PARPi while maximizing anti-cancer effects
(ii) resensitize patients who developed PARPi resistance (iii) switch from an HRP to an HRD status.
PDT has emerged as a new innovative treatment methodology in cancer [10]. This minimally invasive treatment, involves the light-activation of a photosensitive drug (PS) causing a photochemical reaction that releases reactive oxygen species (ROS) thus leading to direct tumor cell death and the modulation biological processes such as the establishment of an anti-tumor immune response thanks to its immunogenic cell death properties [11]. More interestingly, the use of PDT for EOC has already been evaluated in phase I and II clinical trials, but severe morbidity (e.g. digestive perforation, capillary leak syndrome and pleural effusion) and no significant objective complete response were observed, mainly due to the lack of tumor specificity for PS uptake. Based on these studies, precise targeting of peritoneal residual cancer cells by PS is required to enable intraperitoneal PDT for EOC. The cell surface protein, folate receptor alpha (FRα) which is overexpressed in 75% of women suffering from EOC [12–15] in contrast to its highly restricted expression in normal tissues represents an interesting biomolecular target with clinical promising prospect in resistant patients.
We have developed and patented a FRα targeted PS (i.e., Folic Acid conjugated to pyropheophorbide-a (Pyro- a) via a polyethylene glycol (PEG) type spacer, named Pyro-PEG-FA, (patent WO/2019/016397)) for PDT treatment of Peritoneal metastases of OC to specifically target peritoneal residual ovarian cancer cell overexpressing FRα. In previous studies, we have shown that this Pyro-PEG-FA was capable to induce direct human ovarian tumor cells death and enhance the activation of the immune system [16]. In vivo, in a humanized mice model of peritoneal ovarian cancer, this targeted PDT exhibited a decrease of the tumor growth and the induction of an anti-tumor immune response was reflected by an increase in the percentage of innate and adaptative immune cells [17].
Preclinical models of cancer are essential to develop effective treatments, and clinical-relevant models that can capture the inter-patient heterogeneity are even more needed in the era of personalized medicine. Although 2D cell lines are widely used as pre-clinical models and are suitable for numerous functional testing they only poorly reflect the patient tumor heterogeneity [18]. Furthermore, some resistance to PDT has already been observed in 3D tumor models compared to 2D ones [19]. For this reason, the evaluation of a new therapies on relevant model like patient-derived tumor Organoids (PDTO) should not be underestimated to evaluate the potential patient’s response.
PDTO can form self-organized 3D micro-tumors that reflect the initial tumor in terms of histological and molecular characteristics [20]. PDTO can efficiently be derived from ovarian tumor [21–23] and a study comparing an ovarian PDTO model with other preclinical models showed that PDTO recapitulated the patient's response while being more relevant than 2D cell lines and faster and more easily cultured than PDX [24]. PDTOs thus represent models close to the clinic that can be used for functional testing and for research purposes.
Head and neck PDTO have been used for testing in vitro EGFR-targeted PDT [25] and 3D model derived from OC cell line to evalute benzoporphyrine derivative PDT combined to chemotherapy [26], but to our knowledge, no study to date has assessed the value of FR
Photosensitizer. The present study use a PS patented by our research unit (patent number WO/2019/016397). Its chemical structure is shown in Figure 1 and is based on folic acid conjugated to pyropheophorbide-a (Pyro) via a polyethylene glycol (PEG) type spacer. The synthesis and the photophysical properties, has already been described in the patent quoted above and its biological effect in previous in vitro and in vivo studies [16,17].
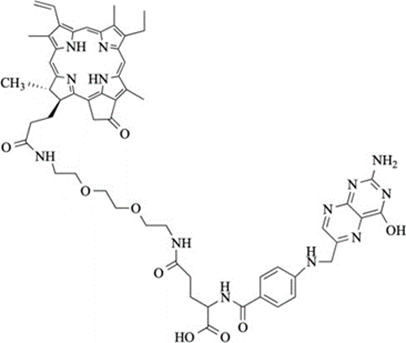
Figure 1: Structure of the Pyro-PEG-FA: folic acid conjugated to pyropheophorbide-a via a polyethy- lene glycol type spacer.
Ovarian Cancer Cell lines
Cell line culture. OC cell lines (OVCAR3 and SKOV3) were ordered from the American Type Culture Collection (ATCC, Manassas, Virginia, USA). SKOV3 cells were cultured in 50% DMEM medium (4.5 g/L D-glucose, L- glutamine, Gibco, Thermo Fisher Scientific, Waltham, MA, USA) and 50% F-12 (Ham’s F-12 Nutrient Mix, Gibco, Thermo Fisher Scientific, Waltham, MA, USA) and OVCAR3 cells were cultured in RPMI-1640 medium (Gibco, Thermo Fisher Scientific, Waltham, MA, USA). The mediums were supplemented with 10% heat inactivated fetal calf serum (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) and with 100 units/mL penicillin and 100 µg/mL streptomycin (Gibco, Thermo Fisher Scientific, Waltham, MA, USA). Cells were maintained in an incubator at 37°C, 5% CO2, and 95% humidity.
Evaluation of the Pyro-PEG-FA incorporation. Adherent cells were cultured on glass coverslips in 12-well plates (Corning, Somerville, MA, USA). 24 h after plating, 9 µM of Pyro-PEG-FA were added to the wells during 24 h. The cell monolayers were washed with PBS (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) and fixed with paraformaldehyde (PFA 4%; Alfa Aesar). After 3 washes, cells were incubated for 1h at RT in PBS containing gelatin (1.2%), glycine (0.2 M) and tween-20 (0.05%) (Sigma-Aldrich, St Louis, MO, USA) for blocking. After washing, the cells were incubated for 1 h with the primary FRα polyclonal antibody (Rabbit/IgG) (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA), diluted (1:400) in PBS overnight at 4°C and after washing, incubated with the secondary antibody Alexa Fluor 555 (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA) (1:300) diluted in PBS for 1h at room temperature (RT). After washing, the nuclei were counterstained with DAPI (Thermo Fisher Scientific, Waltham, MA, USA). Coverslips were mounted in Glycergel (Agilent Dako, C056330-2, Santa Clara, California, USA, USA) and observed using a LSM 710 inverted confocal microscope (Leica, Wetzlar, Germany). Photographic images were resized, organized, and labeled using Fiji-ImageJ software V2.3.0/1.53q.
Cell lines response to PDT. OVCAR3 cell line (10 000 cells) and SKOV3 cell line (5.000 cells) were seeded in a white clear bottom 96-well plates (Corning, Somerville, MA, USA). After 24 h, the medium was replaced by a fresh one containing Pyro-PEG-FA at different concentrations from 0 to 4.5 µM (0, 0.14, 0.28, 0.56, 1.12, 2.25 and 4.5 µM). 24 h later, the medium con-taining PS was changed and replaced by the usual medium of the cell type after two washing steps with PBS. A homogeneous illumination (1 mW/cm2) was then performed at 1.8 J/cm2 with a specific 672 nm laser-based device developed by our research unit [38]. All the experiments were performed in dark conditions. Cancer cell lines viability after PDT was assessed 24 h after the treatment by CellTiterGlo assay according to the manufacturer instruction (CellTiterGlo®, Promega, Madison, WI, USA). The luminescence (RLU) was quantified using a luminometer ClarioStar Plus (BMG Labtech, Champigny sur Marne, France) driven by MARS™ Software v2.06. Viability values were normalized to non-treated control and treatment sensitivity was expressed as the average of three independent replicates expressed in percentage of Normalized Viability.
3D cultures derived from cell lines
3D cultures derived from the OVCAR3 cell line establishment. 3D cultures were derived from the OVCAR3 cell line previously described. 2000 isolated cells were seeded in domes of 20 µL containing 2:3 growth factor- reduced matrigel (Corning® 356231) and 1:3 organoid-specific medium. Specific medium contained 50% Advanced DMEM basal medium (Gibco, Thermo fischer) and 50% prepared medium containing: 10%HA-E- Spondin1-Fc 293 T conditioned medium, 50% L-WRN conditioned medium, N-Acetyl-L-cysteine (1.25 mM), Supplement B27 100X (Thermo Fischer), recombinant mouse EGF (50 ng/ml) (Thermo Fischer), recombinant human FGF-10 (20 ng/ml) (Peprotech), recombinant human FGF-Basic (1 ng/ml), A-83-01 (500nM), Y-27632 dihydrochloride (10 μM) (Stemcell), SB202190 (1 μM) (Gibco, ThermoFischer), Nicotinamide (10 mM) (Sigma- Aldrich), Prostaglandin E2 (1 μM) (SigmaAldrich). 3D cultures were maintained in culture for 15 days at 37°C under 5% CO2 before being transferred to 96-well plates for PDT treatment.
FRα expression. 2000 OVCAR3 cells were seeded in Matrigel domes affixed on a coverslip in a 24-well plate (Corning, Somerville, MA, USA) containing 1 Matrigel dome/well. After 20 days of culture, the medium was removed and incubated with 4% PFA for 20 min at RT then washed with PBS. 3D cultures were permeabilized with a 0.2%saponin solution diluted in PBS (Sigma-Aldrich) for 20 min at RT. The non-specific sites were then saturated with a mixture of 0.2% saponin and 3% BSA (Sigma-Aldrich) for 20 min at RT. Anti-FRα primary antibody (Polyclonal antibody, Rabbit IgG, Invitrogen) was incubated in the same solution at 4°C overnight.
After 3 washes with PBS, the secondary antibody (Alexa Fluor 488 goat anti-rabbit IgG, Invitrogen) was added for 1 h 30 at RT in the dark. Slides were mounted on coverslips using 15 µL of mounting liquid (Vectashiel®, Vector Laboratories) per slide on SuperFROST slides (CML) and observed using a LSM 710 inverted confocal microscope (Leica, Wetzlar, Germany). Photographic images were resized, organized, and labeled using Fiji- ImageJ software V2.3.0/1.53q.
3D cultures derived from the OVCAR3 cell line response to PDT. Organoids were seeded in a white clear bottom 96-well plates (Corning, Somerville, MA, USA). After 24 h, the medium was replaced by a fresh one containing Pyro-PEG-FA at 9 µM. 24 h later, the medium containing Pyro-PEG-FA was changed and replaced by the usual medium after two washing steps with PBS. A homogeneous illumination (1 mW/cm2) was then performed at 3.6 J/cm2 with the same laser-based device as previously described. All the experiments were performed in dark conditions. Organoids viability after PDT was assessed 24 h after the treatment by 3D CellTiterGlo assay according to the manufacturer instruction (CellTiterGlo®, Promega, Madison, WI, USA). The luminescence (RLU) was quantified using a luminometer ClarioStar Plus (BMG Labtech, Champigny sur Marne, France) driven by MARS™ Software v2.06. Treatment sensitivity was expressed as the average of three independent replicates expressed in percentage in RLU.
Patient-derived tumor organoids (PDTO)
Ethical considerations and regulatory aspects. Fresh tumoral tissue from EOC were collected from patients treated at the Comprehensive Cancer Center Francois Baclesse (Unicancer Center, Normandy) by the Biological Resources Center ‘OvaRessources’ (NF-S 96900 quality management, AFNOR No. 2016: 72860.5). The biological collection was declared to the French Ministry of Education, Health and Research (No. DC 2010- 1243). Informed consent form was signed by the patients and was obtained under the agreement of the ethical committee “North-West III” (CPP).
PDTO characteristics. OV-150_A and OV-174_T were derived from patients diagnosed with a HGSOC at a FIGO stage IV with no BRCA mutation. OV-174_T was derived from a tumor tissue extracted from the ovary and OV- 150_A was derived from an ascites sample.
PDTO culture. PDTO were obtained from tumor dissociated cells or filtered ascites as previously described [27]. Cells were collected in organoid basal medium (OBM: Advanced DMEM (Fisher Scientific), 10 UI/mL penicillin, 10 µg/mL streptomycin, 1% GlutaMAX-1 (Fisher Scientific)) and pelleted (2 000 rpm for 5 min). Cells were then resuspended in organoid culture medium (OBM containing B27 (Fischer Scientific, 200 µL/mL), N- Acetyl-L-cysteine (Sigma, 1.25mM), EGF (Miltenyi, 50ng/mL), FGF-10 (Peprotech, 20ng/mL), FGF-basic (Miltenyi, 1ng/mL), A-83-01 (Peprotech, 500nM), Y27632 (Selleckchem, 10µM), SB202190 (Peprotech, 1µM), Nicotinamide (Sigma, 10mM), PGE2 (Sigma, 1µM), Primocin (InvivoGen, 100 µg/mL), Cultrex HA-R-Spondin-1- Fc 293T (AmsBio, 10% V/V) and Cultrex L-WRN (AMS Bio, 50% V/V)). Then, 50µl drops of 1:1 growth factor-reduced BME2 (Bio Techne)/cell suspension containing 10 000 cell per drops were allowed to solidify on prewarmed 24-well suspension culture plates. After polymerization (37°C, 5% CO2, 15 min), each drop was immersed with 500 µL of organoid culture medium. Medium was renewed twice a week and PDTO were passaged every 2-3 weeks: PDTO were collected with cold OBM supplemented with 1% BSA, centrifuged at 200g for 2 min and incubated with TrypLE Express (Gibco, ThermoFischer) for up to 10 min at 37°C. After dissociation, cells were centrifuged at 430g for 5 min, resuspended in organoid culture medium and counted. Then, 50 µl drops of BME2-cell suspension (10 000 cell per drops) were placed in prewarmed 24-well plates. Upon completed gelation, organoid culture medium was added to each well. Plates were then transferred to a humidified 37°C/5% CO2 incubator. PDTO lines were authenticated by comparison of their short tandem repeat (STR) profiles with that of sample of origin (Microsynth).
FRα expression. Tumor tissue and PDTO were fixed in 3% PFA overnight. After embedding PDTO in 2% agarose, tissue and PDTO were dehydrated, paraffin embedded, and sectioned before standard HES staining. Automated immunohistochemistry using a Ventana Discovery XT autostainer (Roche) was performed on 4 µm- thick paraffin sections. Slides were deparaffinized with EZPrep buffer and epitopes were unmasked by 15 min of high-temperature treatment in CC1 EDTA buffer. Sections were incubated for 40 min at 37°C with an anti- FRα (ab221543, Abcam, 1/1500). Secondary antibody (Omnimap Rabbit HRP; Ventana Medical System Inc., Tucson, AZ, USA) was incubated for 16 min at room temperature. Immunodetection performed without the primary antibody was used as control. After washes, the staining was performed with DAB (3, 3'- diaminobenzidine) and sections were counterstained with hematoxylin using Ventana reagents according to the manufacturer's protocol. Stained slides were then digitized using an Aperio ScanScope slide scanner (Aperio Technologies).
PDTO response to Olaparib. Olaparib (Medchemexpress) was diluted in DMSO and stored as a 50 mM stock solution at -80°C. Response of PDTO to treatment was assessed as previously described [28]. When PDTO reached the size of 75-150 µm in diameter, they were collected with cold OBM supplemented with 1% BSA and centrifugated at 200 g for 2 min. Next, the pellet was resuspended in organoid treatment medium (organoid culture medium lacking primocin, Y-27632 and N-acetylcysteine) and counted. PDTO were resuspended in 2% BME2/organoid treatment medium and 200 PDTO per well were seeded in 100 μL volume in a previously coated (1:1 treatment medium/BME2) white clear bottom 96-well plates (Greiner). Thirty minutes later, PDTO were exposed to Olaparib and plates were transferred to a humidified 37°C/5% CO2 incubator. During the treatment, PDTO were monitored using IncuCyte S3 ZOOM (Sartorius). One week later, ATP levels were measured by CellTiter-Glo 3D assay and luminescence was quantified using GloMax Plate Reader (Promega). Viability values were normalized to control and treatment sensitivity was expressed as the average of two independent replicates. To determine status of resistance to Olaparib, results were compared to a collection of other HGSOC PDTO, one reference PDTO was displayed.
Evaluation of the Pyro-PEG-FA incorporation. PDTO were cultured in 6-well plates (Corning, Somerville, MA, USA) and incubated with 9 µM of Pyro-PEG-FA during 24 h. Then, the PDTO were washed with PBS and the nuclei were counterstained with Hoescht (Thermo Fisher Scientific, Waltham, MA, USA). PDTO were then directly observed using a SP8 confocal microscope Gx25, W0.0.95 (Leica, Wetzlar, Germany). Photographic images were resized, organized, and labeled using Fiji-ImageJ software V2.3.0/1.53q.
PDTO response to combination therapy. PDTO were seeded and exposed to Olaparib for one week as described above. During the treatment, plates were kept on a humidified 37°C, 5% CO2 incubator. One week later, medium was removed and PDTO were washed with PBS. PDTO were then exposed to a solution of Pyro- PEG-FA at different concentrations (0, 1.12, 2.25, 3.5 and 4.5 µM). 24 h later, the medium containing Pyro- PEG-FA was changed and replaced by the usual medium after two washing steps. A homogeneous illumination (1 mW/cm2) was then performed at 1.8 J/cm2 with the same laser-based device previously described. 24 h after the illumination, the viability was evaluated using CellTiterGlo 3D assay as described above. Viability values were normalized to control and treatment sensitivity was expressed as the average of two independent replicates.
Statistical Analysis
All results were expressed as the means and standard deviations or standard error of the mean of triplicates of at least three independent experiments. All data were analyzed using the statistical package GraphPad Prism for Windows 3.0.1 (GraphPad, San Diego, CA, USA). The normality of the distributions was assessed using the Shapiro–Wilk test. All quoted p-values are two-sided, with p ≤ 0.05 (*), p ≤ 0.01 (**), p ≤ 0.001 (***), and p ≤ 0.0001 (****) being considered statistically significant for the first and highly significant for the other.
PDT is effective on FRa-expressing ovarian cancer cell lines
The fluorescence properties of the Pyro-PEG-FA were used to assess its uptake by confocal microscopy while the FRα expression by the OC cell lines was confirmed by immunofluorescence (Figure 2). Visual analysis of FRα labeling (in green) confirmed FRα membrane expression by OVCAR3 and SKOV3 cell lines (Figure 2A.1). Regarding the Pyro-PEG-FA uptake (in red), we noticed an incorporation after 24 h of incubation into both the cell lines (Figure 2A.1), which was higher for the OVCAR3 cells compared to SKOV3 cells (Figure 2A.2) thus confirming capacity of the FRapositive cells to internalize the Pyro-PEG-FA.
After validating the Pyro-PEG-FA incorporation into OC cells, we evaluated the efficacy of PDT. In this regard, cells were incubated with various concentrations of PS for 24 h and then illuminated at 1.8 J/cm2. Viability was assessed 24 h post-treatment (Figure 2B). We observed that OC cells were sensitive to PDT through a dose response curve. Moreover, we noticed that OVCAR3 cells were more sensitive to the treatment than SKOV3 cells (EC50 OVCAR3 = 0.174 µM vs EC50 SKOV3 = 0.476 µM). Finally, we demonstrated that none of Pyro-PEG- FA concentration tested was cytotoxic to the cells in dark condition.
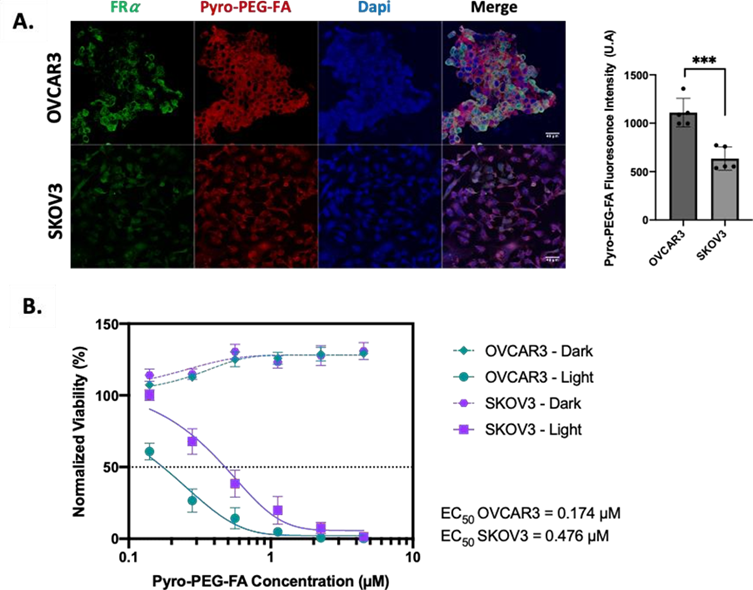
Figure 2: Sensitivity of Ovarian Cancer cell lines to FRα targeted PDT. (A.) Images of Confocal microscopy for FRα expression and Pyro- PEG-FA accumulation in OVCAR3, SKOV3 cell lines cultured with 9 µM of PS for 24 h and observed at Gx40 (left) and semi- quantification of PS fluorescence intensity expressed in U.A (right). (B) Percentage of Normalized Viability for OVCAR3, SKOV3 cell lines 24 h post-PDT treatment. Cells were incubated with increasing concentrations of Pyro-PEG-FA (0.14, 0.28, 0.56, 1.12, 2.25 and 4.5 µM) for 24 h. Illumination was performed with laser set at 672 nm coupled to a specific device delivering 1 mW/cm2 and with a light dose of 1.8 J/cm2. Results are represented as mean ± SEM of 3 independent experiments, expressed in percentage (%) compared to Non- Treated (NT) cells condition. (n=5).
PDT is effective on 3D cultures derived from ovarian cancer cell lines
Next, we wanted to evaluate the PDT potential in more complex 3D model derived from cell lines. First, the FRa expression in 3D cultures derived from the OVCAR3 cell line was evaluated by immunofluorescence (Figure 3A). Fluorescence (in green) reflecting FRa expression confirmed the expression of the receptor by the established 3D cultures after 15 days of culture.
To evaluate the efficacy of PDT, 3D cultures were incubated with 9 µM of Pyro-PEG-FA for 24 h. Based on previous studies [16,17], the organoids were then irradiated at 3.6 J/cm2 with an irradiance of 1mW/cm2. Cell- derived 3D cultures structures were evaluated by IHC at 48h and 72h (Figure 3B) and viability was assessed 1h, 24h, 48h and 72h post-treatment (Figure 3C).
Regarding the morphological aspect of the 3D cultures in PDT condition, the structure looked strongly altered by the treatment 48 h and 72 h post PDT. This is even more interesting, as none of these changes were observed under the other control conditions (Figure 3 B). These observations were confirmed with the viability test as we observed a strong and significant decrease of the viability in the PDT condition from 1 h post PDT (p=0.0049) (Figure 3C). Furthermore, 24 h post-illumination, this decrease was even more significant (p=0.0006) and sustained throughout the assay (until 72 h post-PDT). Concerning the control condition, we found a slight but significant decrease of the viability in the Pyro-PEG-FA and illuminated condition 24 h post treatment (p=0.0158 and p=0.0220 respectively) but this observation was not found anymore 72 h post treatment and remained largely lower than the PDT-condition.
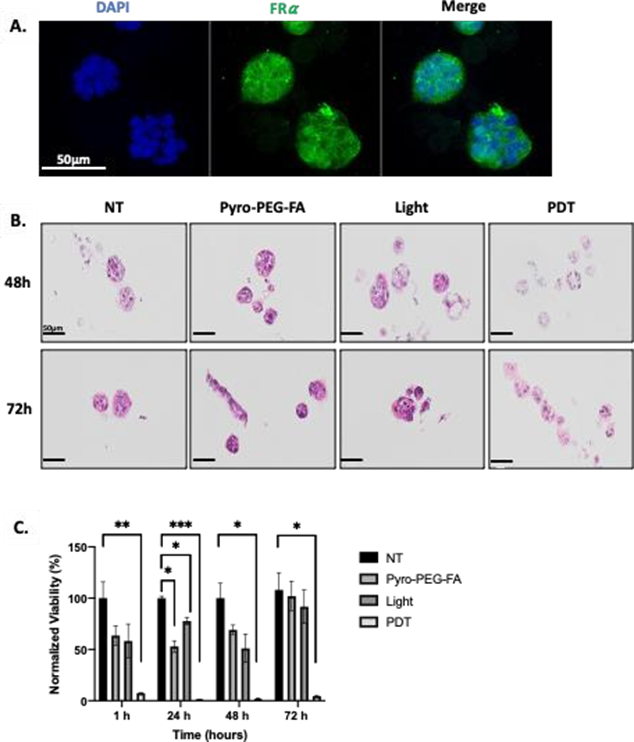
Figure 3: Sensitivity of organoids derived from ovarian cancer cell line to Frα targeted PDT. (A) Images of Confocal microscopy-based FRα expression in organoids derived from OVCAR3 and observed at Gx40 (scale bar = 50 µM). (B) IHC analysis of organoids 48 h and 72 h after PDT. (C) Percentage of normalized viability of the organoids derived from OVCAR3 cell line after PDT treatment. Organoids were treated with 9 µM of PS for 24 h. Illumination was performed with laser set at 672 nm combined to a device delivering 1 mW/cm2 and with a light dose of 3.6 J/cm2. Results are represented as mean ± SEM of 3 independent experiments, expressed in percentage (%) compared to Non-Treated (NT) condition. NT: non-treated; Pyro-PEG-FA: photosensitizer only; Light: illumination only; PDT: illumination in the presence of the Pyro-PEG-FA.
Patient-derived tumor organoids response to PARPi and FRα targeted PDT
Selection of two samples of PDTO resistant to PARPi and eligible to FRα targeted PDT
In order to have a better in vitro model that faithfully represent the patient tumor heterogeneity, we investigated if PDT could open a new therapeutic option for patient resistant to PARPi in ovarian PDTO lines. Firstly, we performed a selection based on two criteria: (i) resistance to PARPi and (ii) expression of the FRα.Based on our first selection criteria, different PDTOs were treated with increasing concentrations of Olaparib and their viability was measured 7 days after treatment (Figure 4A). We intended to use 2 models of PARPi- resistant PDTOs with different degrees of resistance. OV-174_T (derived from a tumor sample) and OV-150_A (derived from an ascites sample) displayed both resistant profile after olaparib treatment in comparison with a reference PDTO with an EC50 of 48.7 µM, 47.1 µM and 11.6 µM respectiveley (Figure 4A right).
Based on the second selection criteria and to assess the efficacy of PDT on PDTOs, we confirmed the expression of the target protein FRα on those two PDTOs by IHC (Figure 4B). We performed an analysis on original tissue and on the resulting PDTOs. We noticed an expression of FRα on original tissue and on PDTO (in brown). Even if the FRα expression is slightly lower in PDTO than in the original tissue it remains important and should be sufficient to perform PDT.
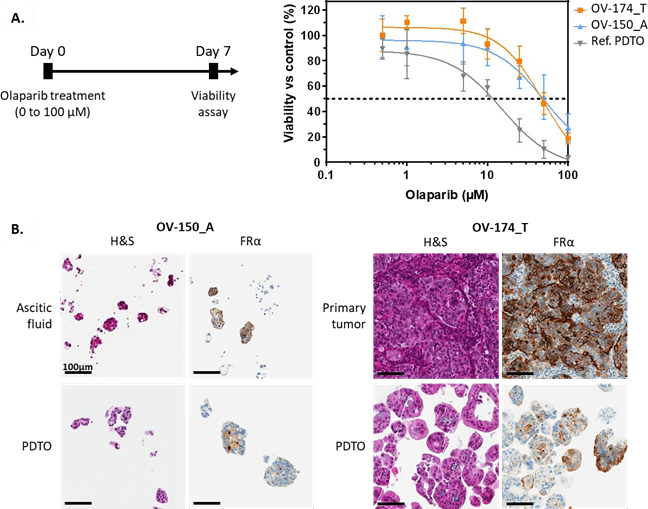
Figure 4: Selection of PDTO resistant to PARPi and eligible to FRα targeted PDT. (A) Percentage of normalized viability of the OV- 150_A, OV-174_T and reference PDTO after 7 days of olaparib treatment. Results are represented as mean ± SD of at least 2 independent experiments. (B) HES and FRα staining of the primary tumor sample and the paired patient-derived tumor organoids for each model selected. Scale = 100 µm.
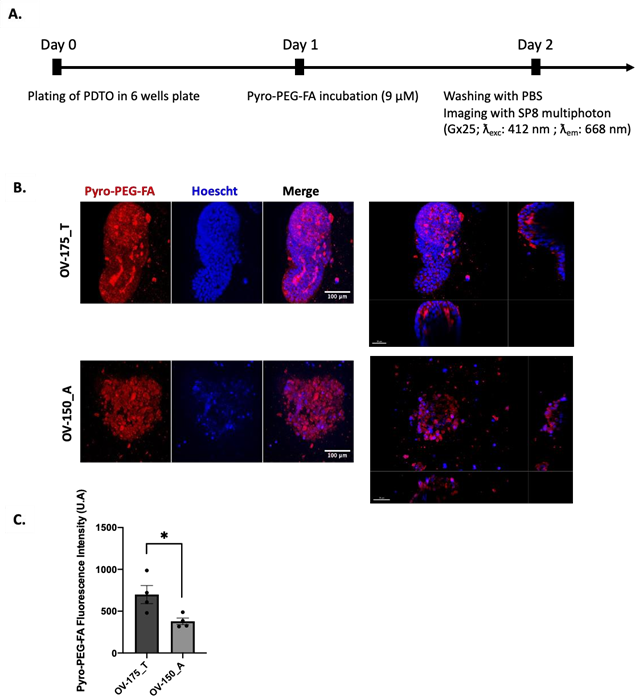
Figure 5: Evaluation of PS uptake within PDTO. (A) Timeline of the treatment (B) Images of Confocal microscopy-based Pyro-PEG-FA uptake within living PDTOs cultured with 9 µM of Pyro-PEG-FA for 24 h and observed at Gx25 (Confocal SP8 multiphoton). Scale bar = 100 µM (left) and 50 µM (right). (C) Semi-quantification of the Pyro-PEG-FA uptake. Histograms are represented as mean +/- SEM of 4 independents experiments.
After confirming the FRα expression, we assessed Pyro-PEG-FA incorporation by living PDTO with imaging thanks to the fluorescence properties of the PS (Figure 5A). We observed a strong red signal reflecting the incorporation of the PS with the two models of PDTO (Figure 5B). The semi-quantification highlighted a greater incorporation for OV-174_T (p=0.0319) (Figure 5C). These results suggest that these PDTOs, resistant to PARPi and expressing FRα, might be sensitive to PDT.
PDTO response to the PARPi-PDT combination treatment
Then, we evaluated the sensitivity of the PDTOs to FRα targeted PDT alone or in combination with Olaparib. Given the high sensitivity of the organoids derived from OVCAR3 cell line to the light dose of 3.6 J/cm2 we reduced the light dose to 1.8 J/cm2 (the same dose as the in vitro model).
For the PDTOs treated with PDT alone, we observed a dose-dependent decrease in cell viability for both types of PDTOs (Figure 6A). We noticed a greater sensitivity of OV-150_A with a decrease of 90 % in viability at a concentration of 3.5 µM, whereas for OV-174_T, we observed a decrease of 20 % at the same concentration.
When PDTOs were treated with PARPi for 7 days and then incubated with the photosensitive drug in dark condition (Figure 6B, PARPi + PS only), we did not notice any effect of the Pyro-PEG-FA since the dose response curve observed was close to the one representing the sensitivity of PDTO to PARPi (Figure 3A). In fact, at the highest concentration of PARPi, the viability of OV-174_T remained at 30% and OV-150_A at 16% confirming the resistance of these models to PARPi. The same observation was noticed when PDTOs were treated with PARPi and illuminated without Pyro-PEG-FA. These results demonstrated that the photosensitive drug alone or the illumination alone do not impact the viability of the organoids when they are treated with PARPi.
Finally, we investigated the effect of the combination PARPi + PDT. PDTOs were first incubated one week with Olaparib before being treated with Pyro-PEG-FA and illuminated (Figure 6C). After bi-therapy treatment, we observed a decrease in PDTOs viability that was dose-dependent on the amount of Pyro-PEG-FA (Figure 6D). However, it appears that PDT alone is sufficient to induce PDTOs death. Indeed, at the highest concentrations of Pyro-PEG-FA (4.5µM in OV-174_T and 3.5 µM in OV-150_A), we observed a decrease in viability that was not dependent on the concentration of Olaparib, resulting in the absence of a dose-response curve. These results tend to show that PDT alone is sufficient to induce PDTOs death and that prior treatment with PARPi does not improve therapeutic efficacy.
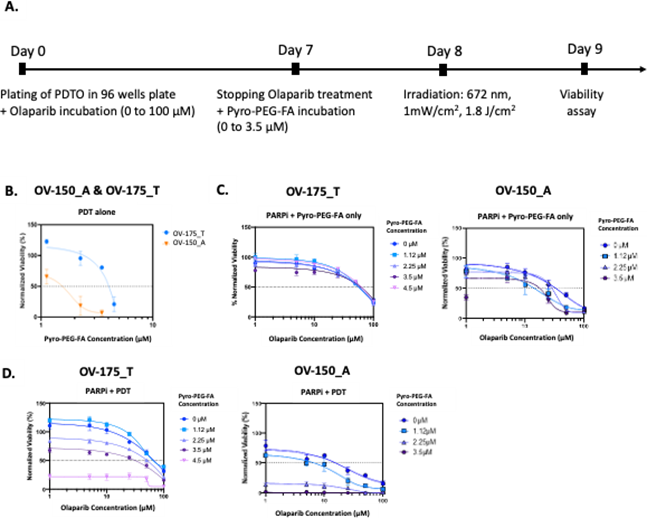
Figure 6: Efficacy of the therapeutic combination PARPi-PDT: (A) Timeline of the combination PARPi – PDT, (B) Percentage of normalized viability of the PDTO after PDT treatment only, (C) after PARPi-Pyro-PEG-FA only treatment, and after (D) the PARPi-PDT treatment. Results are represented as mean ± SD of at least 2 independent experiments.
EOC is one of the most challenging diseases in gynecological oncology, with 80% of advanced cases relapsing after initial therapies [29]. Improved first-line strategy are urgently needed to enhance patient prognosis. In this context, PARPi has emerged to treat ovarian cancers but also various malignancies (e.g., breast, pancreatic, prostate, fallopian and primary peritoneal cancers) [30]. Despite significant improvements in outcomes for patients with HRD thanks to PARPi [29], two major issues remain: (i) PARPi are not devoid of toxicities (ii) resistance to PARPi has been described in both preclinical and clinical settings [5]. Therefore, strategies to reduce side effects associated with PARPi and/or overcome PARPi resistance through combinational therapeutic approaches are urgently needed.
In this context, PDT is an emerging treatment modality in oncology that could be relevant. We previously developed and patented a vectorized PS coupled to Folic Acid in order to target FRα which is overexpressed in 80% of EOC. The effectiveness and the immunogenic properties of this PDT using this photosensitive drug has already been demonstrated in vitro et in vivo in our previous study [16,17].
Preclinical data have demonstrated powerful combination effects of PARPi with PDT [31–33] which could be beneficial for patient. Tanaka et al. were able to show in a gastric cell model that PDT treatment mediated by the photosensitizer talaporfin followed by 48h of exposure to Olaparib produced a synergistic response to the treatments. These results were then confirmed in a xenograft mouse model, with a reduction in tumor volume in the combination condition [32]. Another study in pancreatic cancer demonstrated the strong synergistic effect of PDT mediated by the second-generation photosensitizer chlorin e6 co-delivered with Olaparib in a cell culture model [31]. These last two studies were based on cell culture models, but other teams proposed to use more complex models and were able to test co-delivery of a photosensitizer with the PARPi talazoparib in an ovarian 3D culture model, demonstrating the efficacy of this combination [33]. With the aim of always working with more clinically relevant models, PDTOs were also studied, with the study of EGFR- targeted photodynamic therapy in head and neck PDTOs [34]. Nevertheless, to our knowledge, no study to date has assessed the value of FRa-targeted PDT in combination with PARPi in ovarian PDTO models. Therefore, the present study aimed to evaluate for the first time the efficiency of this FRα targeted PDT on PDTOs selected for their resistance to PARPi. The objectives were to assess the clinical potential of PDT alone for patient resistant to conventional treatments and to evaluate the clinical relevance of the combination of PDT to PARPi.
First, we established dose response curve in a 2D model of ovarian cancer. We observed that the OVCAR3 cell line was more sensitive to PDT than SKOV3 cell line likely due to higher FRα expression in OVCAR3. This difference in PDT sensitivity might also reflect differences in behavior of the primary tumor types from which the cell lines were derived. High-grade serous ovarian carcinoma (HGSOC), from which OVCAR3 is derived, presents at an advanced stage, grows rapidly and spread throughout the peritoneal cavity. In contrast, non- serous tumors (NS) from which SKOV3 is derived, commonly present an early stage and formed large tumor mass. Surprisingly, Amelia Hallas-Potts et al., found that NS cell lines migrated more rapidly than HGSOC and were more invasive which seems counter-intuitive to the clinic. Therefore, even if the conventional 2D cell culture has long been established and allows for a rapid and reliable growth of cancer cells, the major drawback of this model is its inability to reproduce the clinical cancer spectrum [35].
It is essential to develop new models that recapitulate the biological characteristics of tumors and their microenvironment. For this reason, PDTOs can fill the gaps left by cell lines in the testing of new drugs, especially as EOC is a heterogeneous disease and accurate assessment of tumor heterogeneity is important for predicting drug resistance and seeking effective treatments [36]. We have therefore further complicated the 2D model by establishing organoids derived from OVCAR3 cell lines. We increased the light dose at 3.6 J/cm² and showed that these spheroids were very sensitive to PDT. This allowed us to demonstrate the efficacy of PDT in a more complex cellular model.
Nevertheless, PDTOs offer a promising preclinical platform, enabling personalized medicine for EOC patient. PDTOs closely recapitulate heterogeneity of the biological properties of the original tumor and can be used for drug screening and studying resistance mechanisms [37]. We and others have developed different models of ovarian cancer PDTOs derived from patients with different mutational profiles and responses to treatments [21–24], demonstrating the relevance of PDTO as clinical avatars reflecting the real-life heterogeneity.
We selected two PDTO models, OV-150_A and OV-174_T, derived from ovarian ascites and tumor tissue respectively, that both displayed a resistance to olaparib. Firstly, we ensured FRα expression within the original tumors and PDTOs and showed that even lower expression levels in PDTO compared to original tumor were sufficient for the Pyro-PEG-FA to target tumor cells and allow PDT efficacy, resulting in a dose dependent decrease in PDTO viability. Next, we evaluated therapeutic combination of PDT and PARPi and the therapeutic combination did not show a synergistic effect between the two therapies, with the PDT efficacy dominating the whole, reflected in particular at the highest concentrations of Pyro-PEG-FA by the presence of a straight line rather than a dose-response curve. However, we demonstrated the efficacy of PDT in a complex patient- derived models that displayed resistance to PARPi suggesting that PDT could be a promising approach to target tumor cells resistant to conventional treatments.
Finally, we aware of the crucial importance of the tumor microenvironment (immune cells, blood vessels, etc.) in tumor growth and treatment response. As PDT is known to induce robust anti-tumor immune response, develop co-culture system in the presence of immune cells (dendritic cells [38], NK cells [39], macrophages [40] and lymphocytes [41]) to recapitulate inter-cell interactions and communication remains a key point to evaluate immunostimulating properties of PDT.
EOC has a poor prognostic and its management remains a challenge between disseminated malignancies and chemoresistance. In this context, the development of innovative therapeutics strategies as well as more pertinent in vitro models to test them is crucial. In this article we provide evidences of the efficiency of FRα based PDT on different models from immortalized ovarian cell lines to patient-derived tumor organoids. We particularly focused on models displaying a resistance to both carboplatin and PARPi and, even if no synergy was observed when PDT was combined to PARPi, we were able to report for the first time very promising results showing the efficacy of PDT in a context of global resistance to standard first-line chemotherapeutic regimen. PDT should thus open up new treatment prospects for EOC and in the long-term for other cancers overexpressing FRα.
Clearly Auctoresonline and particularly Psychology and Mental Health Care Journal is dedicated to improving health care services for individuals and populations. The editorial boards' ability to efficiently recognize and share the global importance of health literacy with a variety of stakeholders. Auctoresonline publishing platform can be used to facilitate of optimal client-based services and should be added to health care professionals' repertoire of evidence-based health care resources.

Journal of Clinical Cardiology and Cardiovascular Intervention The submission and review process was adequate. However I think that the publication total value should have been enlightened in early fases. Thank you for all.

Journal of Women Health Care and Issues By the present mail, I want to say thank to you and tour colleagues for facilitating my published article. Specially thank you for the peer review process, support from the editorial office. I appreciate positively the quality of your journal.
Journal of Clinical Research and Reports I would be very delighted to submit my testimonial regarding the reviewer board and the editorial office. The reviewer board were accurate and helpful regarding any modifications for my manuscript. And the editorial office were very helpful and supportive in contacting and monitoring with any update and offering help. It was my pleasure to contribute with your promising Journal and I am looking forward for more collaboration.

We would like to thank the Journal of Thoracic Disease and Cardiothoracic Surgery because of the services they provided us for our articles. The peer-review process was done in a very excellent time manner, and the opinions of the reviewers helped us to improve our manuscript further. The editorial office had an outstanding correspondence with us and guided us in many ways. During a hard time of the pandemic that is affecting every one of us tremendously, the editorial office helped us make everything easier for publishing scientific work. Hope for a more scientific relationship with your Journal.

The peer-review process which consisted high quality queries on the paper. I did answer six reviewers’ questions and comments before the paper was accepted. The support from the editorial office is excellent.

Journal of Neuroscience and Neurological Surgery. I had the experience of publishing a research article recently. The whole process was simple from submission to publication. The reviewers made specific and valuable recommendations and corrections that improved the quality of my publication. I strongly recommend this Journal.

Dr. Katarzyna Byczkowska My testimonial covering: "The peer review process is quick and effective. The support from the editorial office is very professional and friendly. Quality of the Clinical Cardiology and Cardiovascular Interventions is scientific and publishes ground-breaking research on cardiology that is useful for other professionals in the field.

Thank you most sincerely, with regard to the support you have given in relation to the reviewing process and the processing of my article entitled "Large Cell Neuroendocrine Carcinoma of The Prostate Gland: A Review and Update" for publication in your esteemed Journal, Journal of Cancer Research and Cellular Therapeutics". The editorial team has been very supportive.

Testimony of Journal of Clinical Otorhinolaryngology: work with your Reviews has been a educational and constructive experience. The editorial office were very helpful and supportive. It was a pleasure to contribute to your Journal.

Dr. Bernard Terkimbi Utoo, I am happy to publish my scientific work in Journal of Women Health Care and Issues (JWHCI). The manuscript submission was seamless and peer review process was top notch. I was amazed that 4 reviewers worked on the manuscript which made it a highly technical, standard and excellent quality paper. I appreciate the format and consideration for the APC as well as the speed of publication. It is my pleasure to continue with this scientific relationship with the esteem JWHCI.

This is an acknowledgment for peer reviewers, editorial board of Journal of Clinical Research and Reports. They show a lot of consideration for us as publishers for our research article “Evaluation of the different factors associated with side effects of COVID-19 vaccination on medical students, Mutah university, Al-Karak, Jordan”, in a very professional and easy way. This journal is one of outstanding medical journal.
Dear Hao Jiang, to Journal of Nutrition and Food Processing We greatly appreciate the efficient, professional and rapid processing of our paper by your team. If there is anything else we should do, please do not hesitate to let us know. On behalf of my co-authors, we would like to express our great appreciation to editor and reviewers.

As an author who has recently published in the journal "Brain and Neurological Disorders". I am delighted to provide a testimonial on the peer review process, editorial office support, and the overall quality of the journal. The peer review process at Brain and Neurological Disorders is rigorous and meticulous, ensuring that only high-quality, evidence-based research is published. The reviewers are experts in their fields, and their comments and suggestions were constructive and helped improve the quality of my manuscript. The review process was timely and efficient, with clear communication from the editorial office at each stage. The support from the editorial office was exceptional throughout the entire process. The editorial staff was responsive, professional, and always willing to help. They provided valuable guidance on formatting, structure, and ethical considerations, making the submission process seamless. Moreover, they kept me informed about the status of my manuscript and provided timely updates, which made the process less stressful. The journal Brain and Neurological Disorders is of the highest quality, with a strong focus on publishing cutting-edge research in the field of neurology. The articles published in this journal are well-researched, rigorously peer-reviewed, and written by experts in the field. The journal maintains high standards, ensuring that readers are provided with the most up-to-date and reliable information on brain and neurological disorders. In conclusion, I had a wonderful experience publishing in Brain and Neurological Disorders. The peer review process was thorough, the editorial office provided exceptional support, and the journal's quality is second to none. I would highly recommend this journal to any researcher working in the field of neurology and brain disorders.

Dear Agrippa Hilda, Journal of Neuroscience and Neurological Surgery, Editorial Coordinator, I trust this message finds you well. I want to extend my appreciation for considering my article for publication in your esteemed journal. I am pleased to provide a testimonial regarding the peer review process and the support received from your editorial office. The peer review process for my paper was carried out in a highly professional and thorough manner. The feedback and comments provided by the authors were constructive and very useful in improving the quality of the manuscript. This rigorous assessment process undoubtedly contributes to the high standards maintained by your journal.

International Journal of Clinical Case Reports and Reviews. I strongly recommend to consider submitting your work to this high-quality journal. The support and availability of the Editorial staff is outstanding and the review process was both efficient and rigorous.

Thank you very much for publishing my Research Article titled “Comparing Treatment Outcome Of Allergic Rhinitis Patients After Using Fluticasone Nasal Spray And Nasal Douching" in the Journal of Clinical Otorhinolaryngology. As Medical Professionals we are immensely benefited from study of various informative Articles and Papers published in this high quality Journal. I look forward to enriching my knowledge by regular study of the Journal and contribute my future work in the field of ENT through the Journal for use by the medical fraternity. The support from the Editorial office was excellent and very prompt. I also welcome the comments received from the readers of my Research Article.

Dear Erica Kelsey, Editorial Coordinator of Cancer Research and Cellular Therapeutics Our team is very satisfied with the processing of our paper by your journal. That was fast, efficient, rigorous, but without unnecessary complications. We appreciated the very short time between the submission of the paper and its publication on line on your site.

I am very glad to say that the peer review process is very successful and fast and support from the Editorial Office. Therefore, I would like to continue our scientific relationship for a long time. And I especially thank you for your kindly attention towards my article. Have a good day!

"We recently published an article entitled “Influence of beta-Cyclodextrins upon the Degradation of Carbofuran Derivatives under Alkaline Conditions" in the Journal of “Pesticides and Biofertilizers” to show that the cyclodextrins protect the carbamates increasing their half-life time in the presence of basic conditions This will be very helpful to understand carbofuran behaviour in the analytical, agro-environmental and food areas. We greatly appreciated the interaction with the editor and the editorial team; we were particularly well accompanied during the course of the revision process, since all various steps towards publication were short and without delay".

I would like to express my gratitude towards you process of article review and submission. I found this to be very fair and expedient. Your follow up has been excellent. I have many publications in national and international journal and your process has been one of the best so far. Keep up the great work.

We are grateful for this opportunity to provide a glowing recommendation to the Journal of Psychiatry and Psychotherapy. We found that the editorial team were very supportive, helpful, kept us abreast of timelines and over all very professional in nature. The peer review process was rigorous, efficient and constructive that really enhanced our article submission. The experience with this journal remains one of our best ever and we look forward to providing future submissions in the near future.

I am very pleased to serve as EBM of the journal, I hope many years of my experience in stem cells can help the journal from one way or another. As we know, stem cells hold great potential for regenerative medicine, which are mostly used to promote the repair response of diseased, dysfunctional or injured tissue using stem cells or their derivatives. I think Stem Cell Research and Therapeutics International is a great platform to publish and share the understanding towards the biology and translational or clinical application of stem cells.

I would like to give my testimony in the support I have got by the peer review process and to support the editorial office where they were of asset to support young author like me to be encouraged to publish their work in your respected journal and globalize and share knowledge across the globe. I really give my great gratitude to your journal and the peer review including the editorial office.

I am delighted to publish our manuscript entitled "A Perspective on Cocaine Induced Stroke - Its Mechanisms and Management" in the Journal of Neuroscience and Neurological Surgery. The peer review process, support from the editorial office, and quality of the journal are excellent. The manuscripts published are of high quality and of excellent scientific value. I recommend this journal very much to colleagues.

Dr.Tania Muñoz, My experience as researcher and author of a review article in The Journal Clinical Cardiology and Interventions has been very enriching and stimulating. The editorial team is excellent, performs its work with absolute responsibility and delivery. They are proactive, dynamic and receptive to all proposals. Supporting at all times the vast universe of authors who choose them as an option for publication. The team of review specialists, members of the editorial board, are brilliant professionals, with remarkable performance in medical research and scientific methodology. Together they form a frontline team that consolidates the JCCI as a magnificent option for the publication and review of high-level medical articles and broad collective interest. I am honored to be able to share my review article and open to receive all your comments.

“The peer review process of JPMHC is quick and effective. Authors are benefited by good and professional reviewers with huge experience in the field of psychology and mental health. The support from the editorial office is very professional. People to contact to are friendly and happy to help and assist any query authors might have. Quality of the Journal is scientific and publishes ground-breaking research on mental health that is useful for other professionals in the field”.

Dear editorial department: On behalf of our team, I hereby certify the reliability and superiority of the International Journal of Clinical Case Reports and Reviews in the peer review process, editorial support, and journal quality. Firstly, the peer review process of the International Journal of Clinical Case Reports and Reviews is rigorous, fair, transparent, fast, and of high quality. The editorial department invites experts from relevant fields as anonymous reviewers to review all submitted manuscripts. These experts have rich academic backgrounds and experience, and can accurately evaluate the academic quality, originality, and suitability of manuscripts. The editorial department is committed to ensuring the rigor of the peer review process, while also making every effort to ensure a fast review cycle to meet the needs of authors and the academic community. Secondly, the editorial team of the International Journal of Clinical Case Reports and Reviews is composed of a group of senior scholars and professionals with rich experience and professional knowledge in related fields. The editorial department is committed to assisting authors in improving their manuscripts, ensuring their academic accuracy, clarity, and completeness. Editors actively collaborate with authors, providing useful suggestions and feedback to promote the improvement and development of the manuscript. We believe that the support of the editorial department is one of the key factors in ensuring the quality of the journal. Finally, the International Journal of Clinical Case Reports and Reviews is renowned for its high- quality articles and strict academic standards. The editorial department is committed to publishing innovative and academically valuable research results to promote the development and progress of related fields. The International Journal of Clinical Case Reports and Reviews is reasonably priced and ensures excellent service and quality ratio, allowing authors to obtain high-level academic publishing opportunities in an affordable manner. I hereby solemnly declare that the International Journal of Clinical Case Reports and Reviews has a high level of credibility and superiority in terms of peer review process, editorial support, reasonable fees, and journal quality. Sincerely, Rui Tao.

Clinical Cardiology and Cardiovascular Interventions I testity the covering of the peer review process, support from the editorial office, and quality of the journal.

Clinical Cardiology and Cardiovascular Interventions, we deeply appreciate the interest shown in our work and its publication. It has been a true pleasure to collaborate with you. The peer review process, as well as the support provided by the editorial office, have been exceptional, and the quality of the journal is very high, which was a determining factor in our decision to publish with you.
The peer reviewers process is quick and effective, the supports from editorial office is excellent, the quality of journal is high. I would like to collabroate with Internatioanl journal of Clinical Case Reports and Reviews journal clinically in the future time.

Clinical Cardiology and Cardiovascular Interventions, I would like to express my sincerest gratitude for the trust placed in our team for the publication in your journal. It has been a true pleasure to collaborate with you on this project. I am pleased to inform you that both the peer review process and the attention from the editorial coordination have been excellent. Your team has worked with dedication and professionalism to ensure that your publication meets the highest standards of quality. We are confident that this collaboration will result in mutual success, and we are eager to see the fruits of this shared effort.

Dear Dr. Jessica Magne, Editorial Coordinator 0f Clinical Cardiology and Cardiovascular Interventions, I hope this message finds you well. I want to express my utmost gratitude for your excellent work and for the dedication and speed in the publication process of my article titled "Navigating Innovation: Qualitative Insights on Using Technology for Health Education in Acute Coronary Syndrome Patients." I am very satisfied with the peer review process, the support from the editorial office, and the quality of the journal. I hope we can maintain our scientific relationship in the long term.
Dear Monica Gissare, - Editorial Coordinator of Nutrition and Food Processing. ¨My testimony with you is truly professional, with a positive response regarding the follow-up of the article and its review, you took into account my qualities and the importance of the topic¨.

Dear Dr. Jessica Magne, Editorial Coordinator 0f Clinical Cardiology and Cardiovascular Interventions, The review process for the article “The Handling of Anti-aggregants and Anticoagulants in the Oncologic Heart Patient Submitted to Surgery” was extremely rigorous and detailed. From the initial submission to the final acceptance, the editorial team at the “Journal of Clinical Cardiology and Cardiovascular Interventions” demonstrated a high level of professionalism and dedication. The reviewers provided constructive and detailed feedback, which was essential for improving the quality of our work. Communication was always clear and efficient, ensuring that all our questions were promptly addressed. The quality of the “Journal of Clinical Cardiology and Cardiovascular Interventions” is undeniable. It is a peer-reviewed, open-access publication dedicated exclusively to disseminating high-quality research in the field of clinical cardiology and cardiovascular interventions. The journal's impact factor is currently under evaluation, and it is indexed in reputable databases, which further reinforces its credibility and relevance in the scientific field. I highly recommend this journal to researchers looking for a reputable platform to publish their studies.
Dear Editorial Coordinator of the Journal of Nutrition and Food Processing! "I would like to thank the Journal of Nutrition and Food Processing for including and publishing my article. The peer review process was very quick, movement and precise. The Editorial Board has done an extremely conscientious job with much help, valuable comments and advices. I find the journal very valuable from a professional point of view, thank you very much for allowing me to be part of it and I would like to participate in the future!”

Dealing with The Journal of Neurology and Neurological Surgery was very smooth and comprehensive. The office staff took time to address my needs and the response from editors and the office was prompt and fair. I certainly hope to publish with this journal again.Their professionalism is apparent and more than satisfactory. Susan Weiner

My Testimonial Covering as fellowing: Lin-Show Chin. The peer reviewers process is quick and effective, the supports from editorial office is excellent, the quality of journal is high. I would like to collabroate with Internatioanl journal of Clinical Case Reports and Reviews.

My experience publishing in Psychology and Mental Health Care was exceptional. The peer review process was rigorous and constructive, with reviewers providing valuable insights that helped enhance the quality of our work. The editorial team was highly supportive and responsive, making the submission process smooth and efficient. The journal's commitment to high standards and academic rigor makes it a respected platform for quality research. I am grateful for the opportunity to publish in such a reputable journal.
My experience publishing in International Journal of Clinical Case Reports and Reviews was exceptional. I Come forth to Provide a Testimonial Covering the Peer Review Process and the editorial office for the Professional and Impartial Evaluation of the Manuscript.

I would like to offer my testimony in the support. I have received through the peer review process and support the editorial office where they are to support young authors like me, encourage them to publish their work in your esteemed journals, and globalize and share knowledge globally. I really appreciate your journal, peer review, and editorial office.
Dear Agrippa Hilda- Editorial Coordinator of Journal of Neuroscience and Neurological Surgery, "The peer review process was very quick and of high quality, which can also be seen in the articles in the journal. The collaboration with the editorial office was very good."

I would like to express my sincere gratitude for the support and efficiency provided by the editorial office throughout the publication process of my article, “Delayed Vulvar Metastases from Rectal Carcinoma: A Case Report.” I greatly appreciate the assistance and guidance I received from your team, which made the entire process smooth and efficient. The peer review process was thorough and constructive, contributing to the overall quality of the final article. I am very grateful for the high level of professionalism and commitment shown by the editorial staff, and I look forward to maintaining a long-term collaboration with the International Journal of Clinical Case Reports and Reviews.
To Dear Erin Aust, I would like to express my heartfelt appreciation for the opportunity to have my work published in this esteemed journal. The entire publication process was smooth and well-organized, and I am extremely satisfied with the final result. The Editorial Team demonstrated the utmost professionalism, providing prompt and insightful feedback throughout the review process. Their clear communication and constructive suggestions were invaluable in enhancing my manuscript, and their meticulous attention to detail and dedication to quality are truly commendable. Additionally, the support from the Editorial Office was exceptional. From the initial submission to the final publication, I was guided through every step of the process with great care and professionalism. The team's responsiveness and assistance made the entire experience both easy and stress-free. I am also deeply impressed by the quality and reputation of the journal. It is an honor to have my research featured in such a respected publication, and I am confident that it will make a meaningful contribution to the field.

"I am grateful for the opportunity of contributing to [International Journal of Clinical Case Reports and Reviews] and for the rigorous review process that enhances the quality of research published in your esteemed journal. I sincerely appreciate the time and effort of your team who have dedicatedly helped me in improvising changes and modifying my manuscript. The insightful comments and constructive feedback provided have been invaluable in refining and strengthening my work".

I thank the ‘Journal of Clinical Research and Reports’ for accepting this article for publication. This is a rigorously peer reviewed journal which is on all major global scientific data bases. I note the review process was prompt, thorough and professionally critical. It gave us an insight into a number of important scientific/statistical issues. The review prompted us to review the relevant literature again and look at the limitations of the study. The peer reviewers were open, clear in the instructions and the editorial team was very prompt in their communication. This journal certainly publishes quality research articles. I would recommend the journal for any future publications.

Dear Jessica Magne, with gratitude for the joint work. Fast process of receiving and processing the submitted scientific materials in “Clinical Cardiology and Cardiovascular Interventions”. High level of competence of the editors with clear and correct recommendations and ideas for enriching the article.

We found the peer review process quick and positive in its input. The support from the editorial officer has been very agile, always with the intention of improving the article and taking into account our subsequent corrections.

My article, titled 'No Way Out of the Smartphone Epidemic Without Considering the Insights of Brain Research,' has been republished in the International Journal of Clinical Case Reports and Reviews. The review process was seamless and professional, with the editors being both friendly and supportive. I am deeply grateful for their efforts.
To Dear Erin Aust – Editorial Coordinator of Journal of General Medicine and Clinical Practice! I declare that I am absolutely satisfied with your work carried out with great competence in following the manuscript during the various stages from its receipt, during the revision process to the final acceptance for publication. Thank Prof. Elvira Farina

Dear Jessica, and the super professional team of the ‘Clinical Cardiology and Cardiovascular Interventions’ I am sincerely grateful to the coordinated work of the journal team for the no problem with the submission of my manuscript: “Cardiometabolic Disorders in A Pregnant Woman with Severe Preeclampsia on the Background of Morbid Obesity (Case Report).” The review process by 5 experts was fast, and the comments were professional, which made it more specific and academic, and the process of publication and presentation of the article was excellent. I recommend that my colleagues publish articles in this journal, and I am interested in further scientific cooperation. Sincerely and best wishes, Dr. Oleg Golyanovskiy.

Dear Ashley Rosa, Editorial Coordinator of the journal - Psychology and Mental Health Care. " The process of obtaining publication of my article in the Psychology and Mental Health Journal was positive in all areas. The peer review process resulted in a number of valuable comments, the editorial process was collaborative and timely, and the quality of this journal has been quickly noticed, resulting in alternative journals contacting me to publish with them." Warm regards, Susan Anne Smith, PhD. Australian Breastfeeding Association.

Dear Jessica Magne, Editorial Coordinator, Clinical Cardiology and Cardiovascular Interventions, Auctores Publishing LLC. I appreciate the journal (JCCI) editorial office support, the entire team leads were always ready to help, not only on technical front but also on thorough process. Also, I should thank dear reviewers’ attention to detail and creative approach to teach me and bring new insights by their comments. Surely, more discussions and introduction of other hemodynamic devices would provide better prevention and management of shock states. Your efforts and dedication in presenting educational materials in this journal are commendable. Best wishes from, Farahnaz Fallahian.
Dear Maria Emerson, Editorial Coordinator, International Journal of Clinical Case Reports and Reviews, Auctores Publishing LLC. I am delighted to have published our manuscript, "Acute Colonic Pseudo-Obstruction (ACPO): A rare but serious complication following caesarean section." I want to thank the editorial team, especially Maria Emerson, for their prompt review of the manuscript, quick responses to queries, and overall support. Yours sincerely Dr. Victor Olagundoye.

Dear Ashley Rosa, Editorial Coordinator, International Journal of Clinical Case Reports and Reviews. Many thanks for publishing this manuscript after I lost confidence the editors were most helpful, more than other journals Best wishes from, Susan Anne Smith, PhD. Australian Breastfeeding Association.

Dear Agrippa Hilda, Editorial Coordinator, Journal of Neuroscience and Neurological Surgery. The entire process including article submission, review, revision, and publication was extremely easy. The journal editor was prompt and helpful, and the reviewers contributed to the quality of the paper. Thank you so much! Eric Nussbaum, MD
Dr Hala Al Shaikh This is to acknowledge that the peer review process for the article ’ A Novel Gnrh1 Gene Mutation in Four Omani Male Siblings, Presentation and Management ’ sent to the International Journal of Clinical Case Reports and Reviews was quick and smooth. The editorial office was prompt with easy communication.

Dear Erin Aust, Editorial Coordinator, Journal of General Medicine and Clinical Practice. We are pleased to share our experience with the “Journal of General Medicine and Clinical Practice”, following the successful publication of our article. The peer review process was thorough and constructive, helping to improve the clarity and quality of the manuscript. We are especially thankful to Ms. Erin Aust, the Editorial Coordinator, for her prompt communication and continuous support throughout the process. Her professionalism ensured a smooth and efficient publication experience. The journal upholds high editorial standards, and we highly recommend it to fellow researchers seeking a credible platform for their work. Best wishes By, Dr. Rakhi Mishra.

Dear Jessica Magne, Editorial Coordinator, Clinical Cardiology and Cardiovascular Interventions, Auctores Publishing LLC. The peer review process of the journal of Clinical Cardiology and Cardiovascular Interventions was excellent and fast, as was the support of the editorial office and the quality of the journal. Kind regards Walter F. Riesen Prof. Dr. Dr. h.c. Walter F. Riesen.

Dear Ashley Rosa, Editorial Coordinator, International Journal of Clinical Case Reports and Reviews, Auctores Publishing LLC. Thank you for publishing our article, Exploring Clozapine's Efficacy in Managing Aggression: A Multiple Single-Case Study in Forensic Psychiatry in the international journal of clinical case reports and reviews. We found the peer review process very professional and efficient. The comments were constructive, and the whole process was efficient. On behalf of the co-authors, I would like to thank you for publishing this article. With regards, Dr. Jelle R. Lettinga.

Dear Clarissa Eric, Editorial Coordinator, Journal of Clinical Case Reports and Studies, I would like to express my deep admiration for the exceptional professionalism demonstrated by your journal. I am thoroughly impressed by the speed of the editorial process, the substantive and insightful reviews, and the meticulous preparation of the manuscript for publication. Additionally, I greatly appreciate the courteous and immediate responses from your editorial office to all my inquiries. Best Regards, Dariusz Ziora

Dear Chrystine Mejia, Editorial Coordinator, Journal of Neurodegeneration and Neurorehabilitation, Auctores Publishing LLC, We would like to thank the editorial team for the smooth and high-quality communication leading up to the publication of our article in the Journal of Neurodegeneration and Neurorehabilitation. The reviewers have extensive knowledge in the field, and their relevant questions helped to add value to our publication. Kind regards, Dr. Ravi Shrivastava.

Dear Clarissa Eric, Editorial Coordinator, Journal of Clinical Case Reports and Studies, Auctores Publishing LLC, USA Office: +1-(302)-520-2644. I would like to express my sincere appreciation for the efficient and professional handling of my case report by the ‘Journal of Clinical Case Reports and Studies’. The peer review process was not only fast but also highly constructive—the reviewers’ comments were clear, relevant, and greatly helped me improve the quality and clarity of my manuscript. I also received excellent support from the editorial office throughout the process. Communication was smooth and timely, and I felt well guided at every stage, from submission to publication. The overall quality and rigor of the journal are truly commendable. I am pleased to have published my work with Journal of Clinical Case Reports and Studies, and I look forward to future opportunities for collaboration. Sincerely, Aline Tollet, UCLouvain.

Dear Ms. Mayra Duenas, Editorial Coordinator, International Journal of Clinical Case Reports and Reviews. “The International Journal of Clinical Case Reports and Reviews represented the “ideal house” to share with the research community a first experience with the use of the Simeox device for speech rehabilitation. High scientific reputation and attractive website communication were first determinants for the selection of this Journal, and the following submission process exceeded expectations: fast but highly professional peer review, great support by the editorial office, elegant graphic layout. Exactly what a dynamic research team - also composed by allied professionals - needs!" From, Chiara Beccaluva, PT - Italy.

Dear Maria Emerson, Editorial Coordinator, we have deeply appreciated the professionalism demonstrated by the International Journal of Clinical Case Reports and Reviews. The reviewers have extensive knowledge of our field and have been very efficient and fast in supporting the process. I am really looking forward to further collaboration. Thanks. Best regards, Dr. Claudio Ligresti