AUCTORES
Globalize your Research
Research Article | DOI: https://doi.org/10.31579/2692-9759/144
1Magdi Yacoub Heart Foundation, Aswan Heart Centre, Egypt.
2Department of cardiovascular medicine, Fayoum University, Egypt.
*Corresponding Author: Omnia Kamel, Magdi Yacoub Heart Foundation, Aswan Heart Centre, Egypt.
Citation: Omnia Kamel, Mostafa El Husseny, Eslam Ahmed, (2024), Tachycardia induced cardiomyopathy as a hidden cause of Heart failure in Pediatrics; A retrospective single-centre study, Cardiology Research and Reports, 6(6); DOI:10.31579/2692-9759/144
Copyright: © 2024, Omnia Kamel. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 09 October 2024 | Accepted: 16 October 2024 | Published: 25 October 2024
Keywords: arrhythmia; pediatrics, tachycardia-Induced cardiomyopathy; heart failure; left ventricular dysfunction; ablation; supraventricular tachycardia, atrial tachycardia
Introduction: Heart failure in pediatrics represents a clinical challenge that requires a comprehensive approach. Early diagnosis and multidisciplinary management are crucial to improve prognosis in such cases. Understanding the unique aspects of HF in pediatrics is essential for effective treatment and care. Unfortunately, a great proportion of pediatric dilated cardiomyopathy cases are classified as idiopathic. Therefore, there is an increasing need to find out the possible hidden causes for cardiomyopathy.
Methods: We retrospectively analyzed our pediatric electrophysiology clinic data since 2019 until 2024. Among our patients,12 pediatric patients diagnosed with tachycardia-Induced cardiomyopathy were included in our study. We analyzed their baseline clinical, electrocardiographic, and cardiac imaging characteristics, management plans as well as their outcomes during follow up visits.
Results: Study population had a mean age of 9 year with 85% of them, had NYHA III heart failure symptoms at the initial presentations. Average duration of tachycardia is 16 month, and mean heart rate of 140 bpm. Incessant atrial arrythmia was the most common presenting arrythmia (54%) followed by PVCs and AV reentrant tachycardia. Mean LVEF was 35% at time of diagnosis. Ablation was the mainstay of management plan of 11 patients with radiofrequency ablation as the standard modality guided with 3D-electro-anatomical mapping. One patient required cryoablation for right atrial appendage isolation. Recovery of ventricular function was achieved in all ablation cohort with no recurrence of arrythmia along follow up.
Conclusions: We highlight TIC as an important underdiagnosed cause for heart failure in pediatrics via our center experience in such cases.
Introduction
Dilated cardiomyopathy (DCM) represents a significant challenge in the pediatric population, with its underlying causes and demographics have been difficult to establish. Despite the profound impact of this condition, particularly as the leading cause for heart transplantation in children, the specifics of its etiology and evolution have proven elusive.[1] Notably, a substantial proportion of pediatric DCM cases, up to 66%, are classified as idiopathic, with the diagnosis typically occurring between 6 and 18 years of age. 2Moreover, a concerning one-third of patients present with severe NYHA class IV symptoms, and the 5-year survival is around 71%. 2-4 this underscores the critical need for deeper insights into the various subtypes of cardiomyopathy (CM) and their unique risk profiles and trajectories.
Intriguingly, a recent classification has emerged that acknowledges the role of tachycardia-induced cardiomyopathy (TIC) as a non-familial, acquired cause of DCM. [5]
While TIC represents a relatively infrequent cause of cardiomyopathy, it is a potentially treatable entity that may be significantly underestimated in clinical practice. In a large retrospective study examining 673 cases of dilated cardiomyopathy, only a single case was attributed to persistent tachycardia as the primary etiology. [6 ]This discrepancy highlights the challenge in accurately identifying TIC, as the arrhythmias are often perceived as a complication of the cardiomyopathy, rather than the underlying driver. The true incidence of TIC may therefore be severely underreported, obscuring the full scope of this clinically important subtype. Elucidating the prevalence and characteristics of TIC, particularly within the pediatric population, is critical to enable timely, targeted interventions that could improve outcomes for affected patients.
While the precise mechanisms underlying the development of TIC remain incompletely understood, several plausible theories have been proposed. Sustained tachycardia may lead to decreased myocardial perfusion, depletion of high-energy phosphates, and alterations in beta-adrenergic receptor signaling and calcium handling, collectively contributing to impaired contractility and remodeling processes. These pathophysiological changes, characterized by neurohormonal over-activation, natriuretic peptide release, inflammation, ventricular dilation, and systolic and diastolic dysfunction, ultimately culminate in the clinical manifestation of TIC. [7-10] Notably, the heightened intracellular calcium stores in infants and young children may exacerbate the vulnerability to TIC.[11]
This retrospective study analyzed the baseline clinical, electrocardiographic, and cardiac imaging characteristics of 12 pediatric patients diagnosed with Tachycardia-Induced Cardiomyopathy and their long-term outcomes. The patients were evaluated and followed up at our center between 2019 and 2024.
Patients with other pre-existing heart diseases and/or possible triggers for their condition were excluded from the study. Clinical management of the patients was provided in accordance with established practice guidelines.
The diagnosis of TIC was made based on the presence of left ventricular (LV) dysfunction and a prior, persistent, or frequent paroxysmal tachycardia, after excluding other potential etiologies. All patients underwent a detailed clinical history, family and drug history assessment, ambulatory electrocardiographic monitoring, echocardiography, and, if necessary, computed tomography coronary angiography (CTCA) to rule out coronary anomalies and cardiac magnetic resonance imaging (CMR) to exclude structural heart diseases.
Following appropriate management, the patients were followed up at 1, 3, 6 and 12 months with advancing imaging modalities and Holter monitoring to ensure complete recovery. The left ventricular ejection fraction (LVEF) during the follow-up period was analyzed after their complete or partial recovery, as well as its association with prognostic factors.
The study population ranged in age from 48 days to 18 years, with a mean age of 9 years, 84% males. Body weights varied from 4 to 57 kilograms. The majority of patients presented with palpitations and dyspnea, while one patient exhibited failure to thrive, another complained of epigastric pain, and finally another presenting symptom was lower limb swelling. At initial presentation, 85% of the patients were in NYHA class III heart failure.
The earliest onset of symptoms was observed during antenatal screening at 5 months of gestation. The average duration of tachycardia was 16 months, with a mean heart rate of 140 beats per minute. All patients were already on at least two antiarrhythmic medications prior to referral, including sotalol (30%), propranolol (46%), amiodarone (38%), verapamil (15%), and bisoprolol (15%).
The most common underlying arrhythmia substrates: incessant atrial tachycardia (54%) (Figure 1), premature ventricular contractions (23%) (Figure 2), accessory pathway-mediated tachycardia (15%), and atrioventricular nodal reentrant tachycardia (8%). The locations of the incessant atrial tachycardias were parahisian (n=2), coronary sinus (n=2), anterior tricuspid annulus (n=1), and right atrial appendage (n=1). The culprits of the PVCs cases were LCC in one case and tricuspid annulus in the other case.
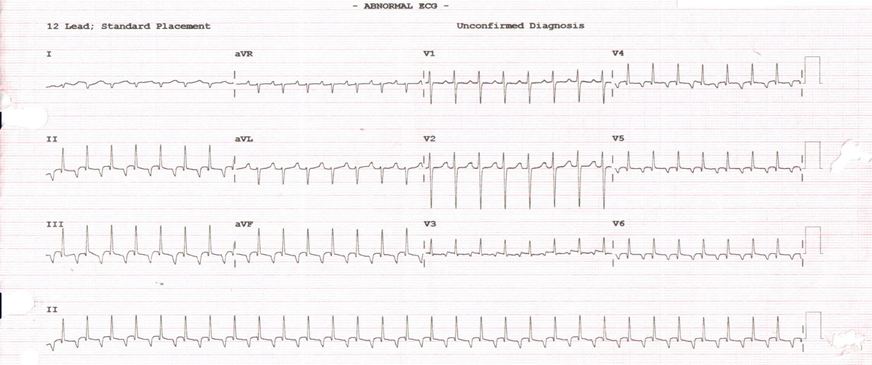
Figure 1: 12 lead ECG shows atrial tachycardia
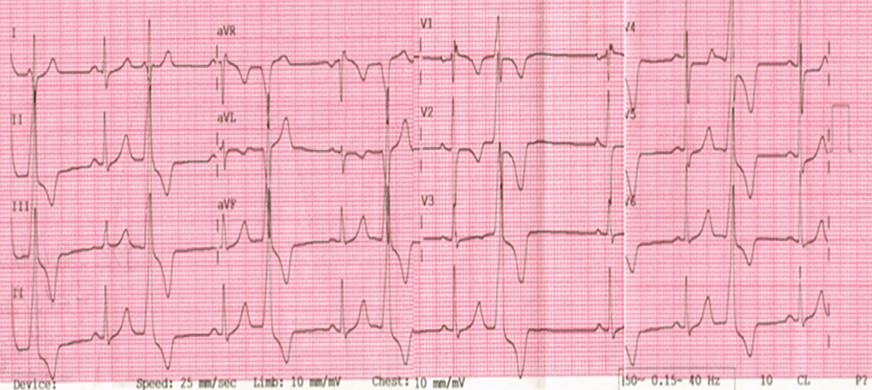
Figure 2: 12 lead ECG shows frequent outflow PVCs
Left ventricular ejection fraction (LVEF) was significantly reduced, ranging from 15% to 48%, with a mean of 35%. In two patients, magnetic resonance imaging (MRI) supported the diagnosis of myocarditis, but these patients did not respond to intravenous immunoglobulin (IVIG) therapy.
All patients, except one who showed non-inducible tachycardia, underwent electrophysiological studies and catheter ablation guided by 3D-
electroanatomical mapping systems (Figure 3). Radiofrequency ablation was the primary modality, with cryoablation used in one patient to ablate the incessant tachycardia originating from the right atrial appendage. Following the ablation procedures, all 12 treated patients remained free of arrhythmia recurrence during the 6-month follow-up period, regardless of the specific underlying arrhythmia.
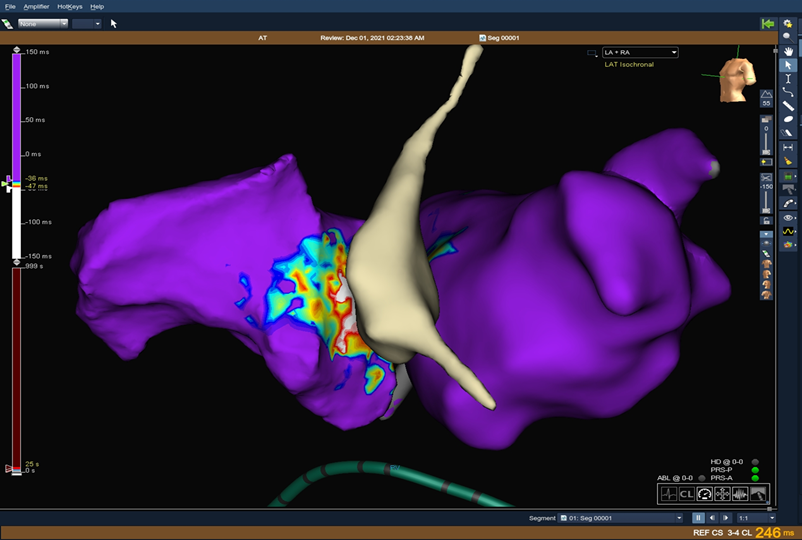
Figure 3: 3D activation map with the earliest atrial activation at parahisian area
Remarkably, complete recovery of LVEF was observed in all patients, with the earliest improvement detected within 2 weeks of successful ablation. In the one patient who was managed medically after failed induction, partial LVEF recovery was noted over 6 months.
Regarding complications, only one patient, weighing 4 kilograms, experienced a right superficial femoral artery thrombosis, which was managed conservatively.
Since its first description in 1949 by Philips and Levine in a young patient who presented with reversible congestive heart failure and AF with rapid
ventricular response [12], TIC has emerged as a potential cause of reversible heart failure with a high burden of morbidity.
Tachycardia-induced cardiomyopathy can arise from various types of tachyarrhythmias, primarily including atrial fibrillation (AF), atrial flutter, incessant supraventricular tachycardia, and frequent premature ventricular complexes (PVCs)[13]. Notably, it may manifest across all age groups—children, adults, and pregnant women—complicating efforts to ascertain its true incidence and prevalence14-16. This variability contributes to the under-recognition of TIC in clinical practice.
The incidence of TIC varies markedly depending on the specific tachyarrhythmia. For instance, it is observed in about 10% of patients with atrial tachycardia (AT) 17, 20-50% among those with incessant AT18, approximately 25% in individuals with atrial flutter19, and up to 50% in patients with AF20. In pediatric populations, AT and permanent junctional reciprocating tachycardia are particularly significant etiological factors for TIC [21].
Additionally, ventricular tachycardia (VT)—especially those originating from outflow tracts or coronary cusps—are among the most common culprits of TIC [22].
Albeit challenging, it is critical to differentiate TIC from other phenotypes of DCM. Spahic et al. conducted a retrospective study examining the characteristics of 18 patients diagnosed with tachycardia-induced cardiomyopathy (TCMP) in comparison to 666 patients with non-ischemic, non-valvular dilated cardiomyopathy (DCM) following episodes of heart failure (HF) decompensation.
Notably, TCMP patients demonstrated a significantly elevated ventricular rate (122 ± 25 bpm) compared to those with DCM (78 ± 21 bpm, p < 0 xss=removed>
In a complementary study, Vera et al. explored the diagnostic role of electrocardiography and cardiac magnetic resonance imaging in patients presenting with HF and concomitant supraventricular tachyarrhythmias. In their analysis of 43 consecutive patients with left ventricular ejection fraction (LVEF) <50>50% during follow-up were classified as TCMP (58%), while those who remained with LVEF <50>
Patients with DCM exhibited wider QRS durations (121.2 ± 26 ms vs. 97.7 ± 17.35 ms; p = 0.003) and higher rates of late gadolinium enhancement (61% vs. 16%; p = 0.004). In contrast, the LVEF in the TCMP group was higher (33.4 ± 11% vs. 26.9 ± 6.4%; p = 0.019). [24].
Over a median follow-up period of 60 months, patients with DCM experienced significantly more frequent hospitalizations for heart failure compared to those with TCMP (44% vs. 0%; p < 0>
The severity of TCMP appears to correlate with the frequency and duration of arrhythmias; research indicates that sustained heart rates of around 110-120 beats per minute may be adequate to initiate the development of cardiomyopathy[ 25]. Notably, in our patient cohort, the average heart rate was around 140 bpm, providing compelling support for the hypothesis that significantly elevated heart rates play a critical role in the pathogenesis of TCMP.
In contrast premature ventricular contractions (PVCs) have been linked to the development of TICM, with the severity of TICM generally correlating with the frequency of ventricular ectopy.26 While frequent PVCs may increase the number of ventricular activations, true tachycardia may not be evident, suggesting additional factors contribute to left ventricular dysfunction beyond mere rate. Mechanistically, persistent electrical activation from PVCs may lead to inefficient mechanical contractions, resulting in dys-synchrony and subsequent LV remodeling.27 Evidence indicates that a PVC burden higher than 20% or an absolute count exceeding 20,000 in 24 hours is associated with reduced LV ejection fraction, though precise thresholds for TIC development remain undefined. [28,29]. Furthermore, PVC QRS duration and origin have emerged as significant predictors of TIC, with longer QRS durations correlating with impaired LVEF and lower chances of recovery.[30].
By analysis of our cohort, we find that PVCs was identified as the culprit behind cardiomyopathy in 23% of the children with a mean QRS duration of 150 msec and in two of them originating from peri-tricuspid annulus.
The vagueness of symptoms in the pediatric population presents a significant obstacle that can delay or even obstruct accurate diagnosis. Unlike adults with TICM who typically report clear symptoms—such as palpitations or shortness of breath—children often exhibit a broad array of nonspecific presentations.
For instance, one particularly challenging case in our cohort involved a 5-year-old child whose primary complaint was persistent epigastric pain, which had lasted for four years. This prolonged symptomatology led to multiple consultations with pediatricians before a referral to a pediatric cardiologist was made, ultimately resulting in a diagnosis of dilated cardiomyopathy.
Complicating the diagnostic process, the patient's electrocardiogram (ECG) revealed a left bundle branch block (LBBB), characterized by a P wave morphology that closely resembled sinus rhythm, save for a positive P wave in V1 that suggested an atrial tachycardia diagnosis (Figure 4). Following an ablative procedure performed within the coronary sinus, the QRS complex normalized, and the patient's ejection fraction returned to the normal range within three months.
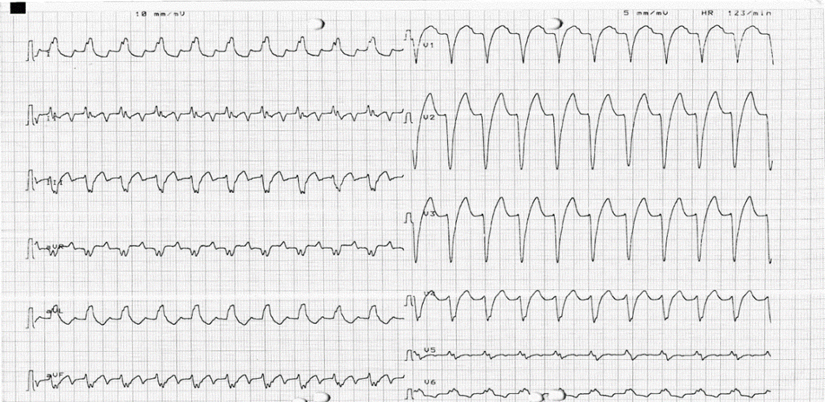
Figure 4: 12 leads ECG shows long RP tachycardia with left bundle morphology with superior axis of P wave
Another case in our study, the youngest patient 48 days old, initially presented with failure to thrive, underscoring the diversity in clinical manifestations of cardiac conditions in the pediatric demographic. These cases highlight the critical need for heightened awareness and thorough evaluation in pediatric patients to facilitate timely and accurate diagnosis of cardiomyopathies.
In TICM, the suppression of the underlying tachyarrhythmia is crucial for restoring left ventricular function. Treatment strategies should prioritize the elimination or management of the culprit tachycardia favorably by radiofrequency catheter ablation (RFCA) or antiarrhythmic medications. Catheter ablation could be challenging especially in small children with low
body weight like one of our cases who was 4Kg, such cases need more considerations regarding vascular accesses, selection of catheters and determining of the endpoint trying to abolish the tachycardia without causing harm (Figure 5 A&B). Evidence from clinical studies suggests that significant improvements in LV systolic function can typically be observed within a month after initiating treatment for tachyarrhythmia. Full recovery often occurs within 2 to 3 months, although it rarely exceeds 6 months.11,31 Importantly, initial management of T-CMP must also involve optimizing medical therapy for heart failure, including the use of beta-blockers, angiotensin-converting enzyme inhibitors, diuretics, and aldosterone blockers, thereby promoting reverse remodeling of the heart.[11]
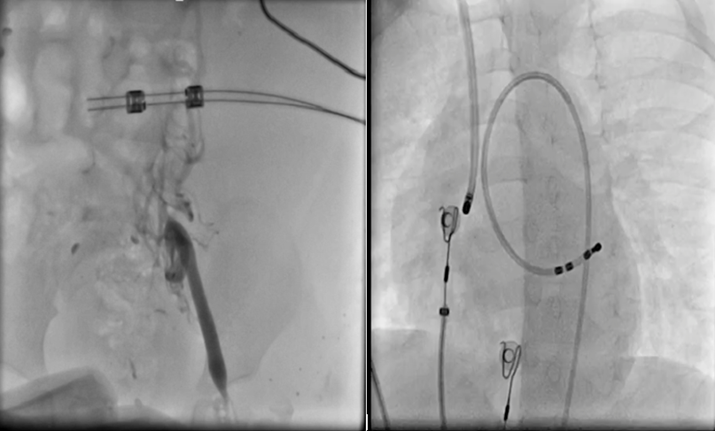
Figure 5 A&B: A. shows injection of totally occluded left femoral vein, B. insertion of ablation catheter via right jugular vein
In alignment with previous findings, an in-depth analysis of our cohort demonstrated that the mean time to recovery of left ventricular ejection fraction was approximately 7 weeks, independent of the duration before
initiating appropriate management. Remarkably, all patients achieved complete recovery except for one individual who remained on medical
treatment due to non-inducibility in the cath lab. The range for full recovery spanned from as little as one week to a maximum of six months. Notably, patients presenting with lower baseline LVEF required a longer duration for complete recovery, highlighting the relationship between initial ventricular function and the time needed for normalization. These observations underscore the variability in recovery trajectories and the importance of early intervention in optimizing outcomes for patients with tachycardia-related cardiomyopathy.
An international multi-centre study conducted by Jeremy. P. Moore and his colleagues involving 81 pediatric patients revealed critical insights into recovery patterns and predictors of outcome in T-CMP. The median age was 4 years, with a baseline LV ejection fraction (LVEF) of 28%. The predominant arrhythmias noted were ectopic atrial tachycardia (59%), permanent junctional reciprocating tachycardia (23%), and ventricular tachycardia (7%). The median recovery times were 51 days for LVEF and 71 days for LV end-diastolic dimension (LVEDD). Multivariate analysis identified several predictors of recovery, including age, tachycardia rate, and baseline LVEF, underscoring the multifaceted nature of prognosis in T-CMP. For LV size normalization, baseline LVEDD proved significant. These findings highlight the importance of timely intervention and comprehensive management strategies in improving outcomes for children with T-CMP.32
Tachycardia-Induced Cardiomyopathy (TICM) has emerged as an often underrecognized culprit behind DCM in pediatric population with a high morbidity and mortality. As we navigate the complexities of this condition, it becomes evident that timely intervention is not just beneficial but essential. The remarkable improvements observed post-ablation highlight the urgency of distinguishing TICM from other forms of dilated cardiomyopathy. As we refine our diagnostic strategies, we must enhance awareness among pediatric healthcare providers on having high index of suspicion for the diagnosis. Every delayed diagnosis not only extends the suffering of our patients but also obscures the potential for full recovery.
Informed consent was obtained from all subjects involved in the study (patients’ parents).
Written informed consent was obtained from the patient parents for the publication of this study and accompanying images.
Conflict of interest: The authors declare that this research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Authors’ contributions: All authors contributed equally.
Funding: No funding was obtained for this study.
Acknowledgements: None