AUCTORES
Globalize your Research
Research Article | DOI: https://doi.org/10.31579/2688-7517/197
Faculdade Ciências Médicas de Minas Gerais
*Corresponding Author: : Thiago Henrique Caldeira de Oliveira, Faculdade Ciências Médicas de Minas Gerais, Alameda Ezequiel Dias, 275 - Centro, Belo Horizonte - MG, 30130-110.
Citation: Caldeira De Oliveira TH, Neves Gonçalves GK, (2025), Effect of Ovariectomy and High-fat Diet on the Expression of Estrogen Receptors and Adipose Tissue Metabolism in Wistar Rats, J. Pharmaceutics and Pharmacology Research, 8(1); DOI:10.31579/2688-7517/197
Copyright: © 2025, Thiago Henrique Caldeira de Oliveira. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 29 May 2024 | Accepted: 01 June 2024 | Published: 13 January 2025
Keywords: adipose tissue; post-menopause; estrogen
Background: This study examines the rising prevalence of obesity in postmenopausal women and its link to hypertension and endothelial dysfunction. Visceral fat poses the highest risk for cardiovascular disease and type 2 diabetes. Estrogen regulates adipose tissue, and its absence post-menopause is linked to metabolic issues.
Aim: The study aimed to determine lipolytic activity in various adipose tissues of high-fat diet-fed ovariectomized Wistar rats, analyze estrogen receptor expression, and perform histological and morphometric analyses of these tissues.
Materials and Methods: 12-week-old female wistar rats were used. The rats were ovariectomized (O) or sham operated (S). Four weeks later, the animals were divided into groups: ovariectomized with high-fat diet (OF), sham-operated with high-fat diet (SF), ovariectomized with control diet (OC) or sham-operated with control diet as the control group (SC). After 24 weeks of consuming the diets, the rats were sacrificed and the mesenteric, parametrial and retroperitoneal adipose tissue deposits were removed, weighed and immediately frozen. Polymerase chain reaction (PCR) was performed to analyze the expression of estrogen receptors alpha (ER-α) and (ER-β) in adipose tissues, lipolysis assay and histological analysis thereof.
Results: The results demonstrated that both the high-fat diet and ovariectomy resulted in increased body weight and adiposity. There was hypertrophy of adipocytes in certain fat deposits. Estrogen replacement therapy was able to modulate lipolytic activity in different adipose depots, with different responses in relation to estrogen receptors. Estrogen receptor expression varied between fat depots and was influenced by diet and ovariectomy. Mesenteric adipose tissue showed greater sensitivity to estrogen compared with parametrial adipose tissue. Estrogen administration increased lipolytic activity in some fat depots, while reducing it in others.
Conclusions: The expression of estrogen receptors is dependent not only on hormonal status, but also on the location of adipose tissue and diet, which may explain the distinct actions of estrogen both on the metabolism of adipose tissue and on the production of adipokines by them.
The prevalence of obesity has increased in recent decades, particularly among postmenopausal women. Obesity is a recognized risk factor for the development of hypertension, often linked with endothelial dysfunction [1-3]. Postmenopause is associated with the development of central obesity, which increases the risk of hypertension, elevated blood lipid levels, insulin resistance, and other cardiovascular diseases [4]. The distribution of adipose tissue has a significant impact on the risks associated with obesity. While visceral fat, which accumulates around the internal organs in the abdominal cavity, is associated with an increased risk of cardiovascular diseases, type 2 diabetes, and metabolic syndrome, subcutaneous fat, which accumulates just below the skin, has a lesser impact on metabolic health [5]. It is suggested that women in the premenopausal stage are protected from these deleterious effects due to the estrogen hormone produced by the ovaries [6].
Estrogen plays a crucial role in regulating the development and storage of adipose tissue in females. Estrogens encompass a group of compounds, comprising estrone, estradiol, and estriol [7]. Estradiol, in particular, can be subdivided into 17 alpha-estradiol (17a-E2) and 17 beta-estradiol (17b-E2) [8]. Among the four types of estrogen, 17β-estradiol, the most potent, exerts its influence through activation of intracellular estrogen receptors known as ER-α and ER-β [9]. After menopause, women tend to increase their adipose mass, largely due to low levels of 17β-estradiol, which may contribute to the development of various chronic diseases, including hypertension. Moreover, Estrogen deprivation has been associated with an increased risk of obesity and metabolic complications, which can be partially reversed by hormone replacement therapy [10].
Despite the epidemiological evidence, it is crucial to conduct additional studies to elucidate the underlying mechanisms by which estrogen influences adipose tissue and the cardiovascular system. Furthermore, it has recently been observed that adipose tissue deposits manifest distinct responses and secretions, varying according to their location in the body [11-13].
The aim of this study was to determine the lipolytic activity in the parametrial, retroperitoneal and mesenteric adipose tissues of ovariectomized Wistar rats submitted to a high-fat diet. In addition, to analyze the expression of estrogen receptors in different types of adipose tissue in animals, as well as histologically and morphometrically analyzing the retroperitoneal, mesenteric and parametrial adipose tissue deposits of ovariectomized Wistar rats fed a high-fat diet.
2.1 Animals
The study was approved by the “Ethics Committee in Animal Experimentation of the Estadual University of Feira de Santana” and carried out according to the recommendations of the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Female Wistar rats weighting 150–200 g was obtained from ICS-UFBA (Salvador, BA, Brazil). The animals (6 per cage) were maintained under controlled light and temperature conditions (lights on from 07:00 to 19:00 h, 23±3 °C) and had free access to tap water and a standard rat chow (AIN 93; Pragsoluções, Brazil). The animals were ovariectomized or sham-operated using ketamine 50 mg/kg and xylazine 5 mg/kg (im). The animals received a prophylactic antibiotic injection (pentabiotic, Fontoura-Wyeth, 0.4 ml/animal/im) [14]. There were no specific exclusion criteria for this study. All animals that meet the inclusion criteria are eligible to participate
2.2 Diet Protocol
After 1-week acclimation, rats were randomly divided into four groups: ovariectomized with high-fat diet (OF, n=6), sham-operated with high-fat diet (SF, n=6), ovariectomized with control diet (OC, n=6) or sham-operated with control diet as the control group (SC, n=6) [14]. Diets were manufactured by Pragsoluções (São Paulo, SP, Brazil) [15]. Body weight and blood pressure were measured weekly for 24 weeks. Then, estrogen replacement therapy was started. The treated groups received 17β-estradiol (Benzoginoestril; SARSA- Hoechst Marion Roussel, 5mg/mL ampoules), diluted in corn oil at a dose of 5µg/0.1ml per 100g of body weight of benzoginoestril, for fourteen consecutive days. Control animals received 0.1mL of corn oil per 100g of body weight during the same period. The rats were killed by decapitation and the mesenteric, parametrial and retroperitoneal adipose tissue depots were removed, weighed and used for further analysis of gene expression of estrogen receptors as well as histological analysis. Part of the tissues was used immediately for the lipolysis assay [16].
2.3 Adipocyte isolation
Adipocytes were isolated from parametrial, retroperitoneal and mesenteric adipose tissues. After digestion with collagenase, cells were filtered and washed three times with 30mmol/L HEPES buffer (pH 7.4) containing 137mmol/L NaCl, 5mmol/L KCl, 4.2mmol/L NaHCO3, 1.3mmol/L CaCl2, MgCl2 0.5mmol/L, MgSO4 0.5mmol/L, KH2PO4. Then, the cells were resuspended in a proportion of 1g of tissue to 20mL of buffer, so that each mL of medium contained 50mg of cells, which were used for lipolysis studies, measuring cell number and diameter [17].
2.4 Lipolysis assay
Adipocytes were incubated under basal conditions or with isoproterenol (0.1 µM) for 90 minutes. Adipocytes from animals treated with vehicle and 17β-estradiol were used in order to characterize their participation in the process. At the end of the incubation period, an aliquot of the infranatant was collected and the glycerol content analyzed. The glycerol released in the lipolysis incubation medium was quantified by an enzymatic kit (ANALISA, Lagoa Santa, MG) [17, 18].
2.5 Diameter and number of cells
The adipocyte suspension was analyzed in a Newbauer chamber, with the number of cells measured in the four outer quadrants of the chamber, and the cells in the inner quadrant used to analyze cell diameter. To calculate the number of cells, the following formula was used: No cells/mL = (No of cells counted) ÷ (No of quadrants counted) X 104 X dilution factor [19].
2.6 mRNA extraction
Tissues were removed and frozen in liquid nitrogen. Total RNA was extracted from adipose tissues according to the thiocyonate-phenol-chloroform method. Total RNA was extracted using Brazol@ reagent at a proportion of 1ml/100mg of tissue. The RNA concentration was measured in a spectrophotometer at an absorbance of 260nm [16].
2.7 Reverse Transcription
The cDNA was obtained through retro-transcription (RT) from the mRNA, using oligonucleotides complementary to the poly-A tail of the mRNA. For reverse transcription, 4ul of specific buffer (10x), 3uL of dNTP (10mM), 4uL of oligo dt primer and 0.2uL of Moloney Murine Leukemia Virus (MMLV) reverse transcriptase enzyme were added to 1µg of total RNA. The solution was then kept at 40°C for 60 minutes for c-DNA synthesis.
2.8 Polymerase chain reaction (PCR)
PCR was performed to amplify the c-DNA for ER-α and ER-β. To amplify ER-α, the following primers were used: sense primer with sequence TAAGAACCGGAGGAACAGTTG, and antisense primer with sequence TCATGCGGAATCGACTTG (product with 623 base pairs); To amplify ER-β, the following were used: sense primer with sequence GCTCCTCTATGCAGAACCTCAAA, and antisense primer with sequence CAGAAGTGAGCATCCCTCTTTG (product with 159 base pairs). PCR was performed using 20µl of MIX solution containing 2.2µl of specific buffer, 1µl dNTP (4mM), 0.2µl of each primer, 1µl of cDNA, 0.1µl of Taq polymerase and 15.30µl of water. The reaction products were analyzed by electrophoresis on a 1.5% agarose gel. The gel was developed with ethidium bromide (1µg/ml), visualized under ultraviolet light and digitalized. The bands were quantified using the ImageQuant 5 program. β-Actin was amplified as an internal control for each PCR using previously described specific primers. The results refer to three PCRs for each sample [16].
2.9 Morphological and histological analysis
Histological sections of adipose tissue measuring 5 micrometers (µm) thick were evaluated. These sections were subjected to hematoxylin-eosin staining for quantitative tissue analysis. Morphometric measurements were carried out using an interactive computerized image analysis system (Image Pro Plus 4.1 imaging software from Media Cybernetics, Silver Spring, MD).
2.10 Data analysis
Data were expressed as mean ± SEM and analyzed using GraphPad software. Data were compared using one-way ANOVA followed by Newman–Keuls post-hoc test or two-way ANOVA followed by Bonferroni post-test. Correlations were calculated using two-tailed Pearson's correlation coefficients. Data was considered statistically significant at the pb0.05 level.
The initial results demonstrate that both high-fat diet and ovariectomy, alone, resulted in an increase in body weight. Furthermore, the combination of a high-fat diet with ovariectomy resulted in a significant increase in body weight, which was also reflected in the different deposits of adipose tissue (Figure 1A). Ovariectomy resulted in an increase in parametrial fat deposition, while the high-fat diet caused an increase in parametrial and mesenteric fat deposits, compared to control animals. However, neither diet nor ovariectomy influenced the weight of the retroperitoneal fat deposit (Figure 1B).
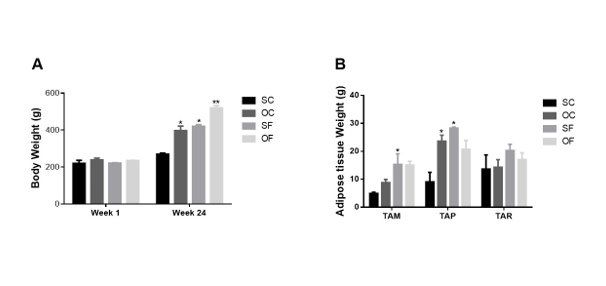
Figure 1: Comparison of weight gain (A) and visceral adipose tissue between animals fed a control and high-fat diet (B) (*p< 0>
The figure 2 shows fat cell morphology after 24 weeks of diet. Control diet animals had uniformly sized adipocytes across all deposits. However, ovariectomized rats on a high-fat diet (OF) exhibited hypertrophic adipocytes in parametrial and mesenteric tissues without increased vasculature, while retroperitoneal tissue showed no significant changes.
The morphological changes in fat distribution induced by a high-fat diet were accompanied by variations in the histological structure of adipose tissue. A high-fat diet caused changes in adipose tissue structure, with the OF group showing increased adipocyte diameter in mesenteric and parametrial tissues, but not in retroperitoneal tissue (Figure 3A). Histological analysis revealed larger adipocytes in mesenteric tissue compared to retroperitoneal and parametrial tissues. Estradiol treatment reduced cell diameter in mesenteric tissue but did not affect parametrial or retroperitoneal adipocytes (Figure 3B).
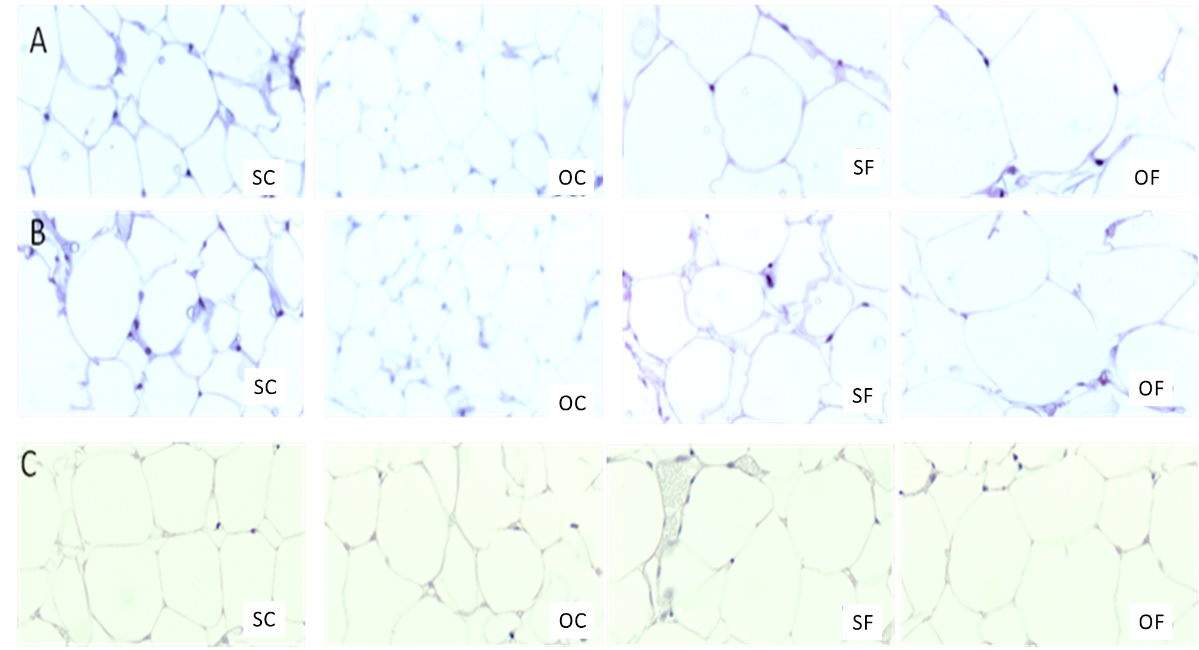
Figure 2: Micrograph of adipose tissue sections from the mesenteric (A), parametrial (B) and retroperitoneal (C) depots of SC, OC, SF and OF rats. Adipose tissue sections from different areas of all deposits were fixed and stained with hematoxylin and eosin. Micrographs were taken at 10x magnitude. Representative sections of each deposit are shown. Micrographs of the parametrial and mesenteric adipose tissue depots of OF rats show hypertrophic adipocytes without evidence of an increase in tissue vasculature. Retroperitoneal adipose tissue does not show any significant changes between groups.
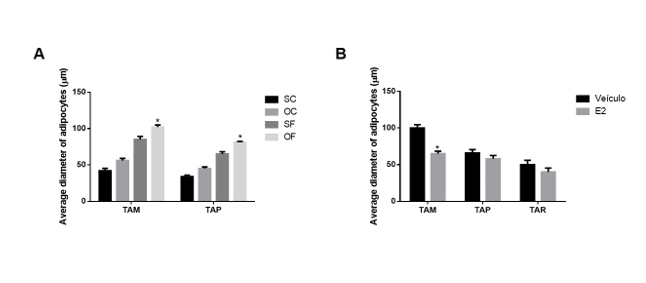
Figure 3: Diameter of adipocytes from the mesenteric (TAM) and parametrial (TAP) adipose tissues of ovariectomized wistar rats fed for 24 weeks with a high-fat diet. * p<0>
A lipolysis assay quantified glycerol release. Isoproterenol, an adrenergic agonist, increased lipolytic activity in all adipose tissues. Retroperitoneal tissue showed higher basal and isoproterenol-stimulated lipolysis. Pretreatment with 17β-estradiol for 14 days reduced lipolysis in retroperitoneal tissue but increased it in parametrial and mesenteric tissues, especially with isoproterenol (Figure 4).
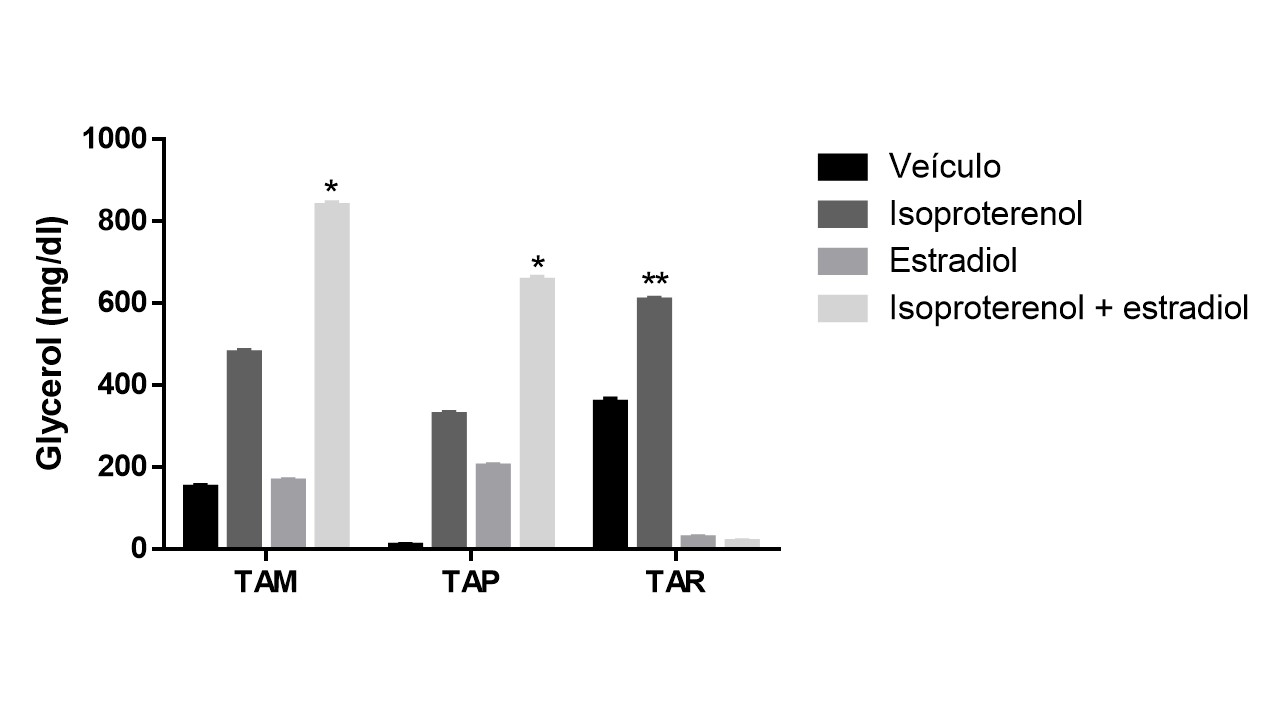
Figure 4: Lipolysis assay of adipocytes isolated from mesenteric (TAM), parametrial (TAP) and retroperitoneal (TAR) adipose tissues of ovariectomized Wistar rats fed a high-fat diet. The results are expressed in mg/dl of glycerol released into the medium under basal conditions or stimulated by isoproterenol. The results refer to the “pool” of adipocytes from five rats treated with vehicle (v) or five rats treated with estradiol (E) for 14 days. *p< 0>
To study the mechanisms involved in the different responses of adipose tissue depots, the gene expression of estrogen receptors was analyzed. Parametrial adipose tissue in control rats showed greater ER-β expression compared to ER-α (Figure 5A), a difference not seen in mesenteric tissue (Figure 5B). After 24 weeks of a high-fat diet, ER-α and ER-β expression in parametrial tissue decreased. Ovariectomy also reduced ER-β expression in this tissue. In contrast, mesenteric tissue showed increased estrogen receptor expression with a high-fat diet, both alone and with ovariectomy.
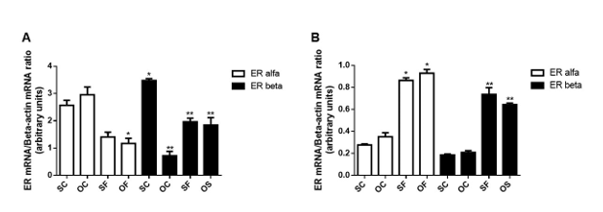
Figure 5: Gene expression of alpha and beta estrogen receptor (ER) in parametrial adipose tissue (TAP) (A) and mesenteric adipose tissue (TAM) (B) of ovariectomized wistar rats fed for 24 weeks with a high-fat diet (OF). * p<0>
Estrogens are considered important regulators of development and lipid deposition in adipose tissue, especially in females. For instance, males typically exhibit a greater propensity for accumulating visceral adiposity, commonly referred to as the "apple shape," heightening the risk of metabolic disorders and cardiometabolic diseases. Conversely, females tend to accrue subcutaneous adipose tissue, providing metabolic protective advantages [20].
To elucidate estrogen's role, estrogen replacement therapy was administered and adipocytes analyzed. Mesenteric adipose tissue, which had the greatest hypertrophy, responded best to therapy, showing a significant reduction in adipocyte diameter after 17β-estradiol treatment.
These data are in line with others found in the literature, which demonstrated that 17β-estradiol decreases cell diameter [21-23]. Furthermore, estradiol replacement therapy in both rodents and humans reverses obesity by reducing visceral fat mass, thereby enhancing metabolic fitness [24, 25]. Increasing evidence from studies conducted in humans and rodents suggests that higher levels of estrogen promote the expansion of subcutaneous adipose tissue while inhibiting the growth of visceral adipose tissue [7]. On the other hand, lower levels of estrogen by menopause or ovariectomy increases the risk of developing obesity, type 2 diabetes and cardiovascular disease [26].
The effects of estrogen on lipid deposition can be attributed to its inhibitory activity on the enzyme lipoprotein lipase (LPL), which regulates lipid uptake by adipocytes. LPL is a crucial enzyme in lipid metabolism. Its main function is to catalyze the hydrolysis of triglycerides present in chylomicrons and very low-density lipoproteins (VLDL), releasing free fatty acids to be absorbed and stored in adipose tissues or used as a source of energy by peripheral tissues [27]. In this regard, when estrogen levels decrease, an increase in LPL activity may occur. Therefore, an increase in LPL activity during menopause may contribute to a greater accumulation of visceral fat and, consequently, increase the risk of developing conditions related to metabolic health [28]. On the other hand, studies demonstrate that estrogen treatment decreases lipid deposition by decreasing LPL activity [29]. Moreover, estrogen markedly decreased the amounts of fat accumulation and LPL mRNA as well as triglyceride accumulation in genetically manipulated 3T3-L1 adipocytes stably expressing the estrogen receptor [30].
Adipose mass reflects both the number and size of the adipocyte, the latter being direct evidence of the stored triacylglycerol content. Thus, the reduction in the mass of parametrial and mesenteric adipose tissue in the OF group treated with estradiol coincides with the reduction in the average diameter of the adipocyte, suggesting that the effect of estradiol in this tissue is mediated, to a large extent, by the effects of this hormone on the adipocyte size. In this regard, studies demonstrate that 17β-Estradiol decreases the intracellular triacylglycerol levels. Moreover, this effect is abolished by ERα antagonist but not ERβ antagonist [31, 32].
The lipolysis assay shows varied tissue responses. Estradiol enhances isoproterenol's effects on mesenteric and parametrial fat, but in parametrial tissue, it acts similarly to isoproterenol alone. In retroperitoneal tissue, estradiol inhibits lipolysis both alone and with isoproterenol. Thus, estradiol increases basal glycerol release from parametrial and mesenteric tissues in rats on a high-fat diet. Similar results were observed in female wistar rats during diestrus, as well as in in situ microdialysis studies performed in ovariectomized females after estrogen administration [9, 33]. These results implicate estrogens as modulators of basal and stimulated lipolytic responses in rat visceral fat in vivo.
It was also observed that adrenergic stimulation by isoproterenol was not influenced differently by hormone replacement in retroperitoneal deposits, corroborating results obtained in women, in whom estradiol replacement therapy did not alter the lipolytic activity stimulated by noradrenaline in subcutaneous adipocytes [34]. These data show that retroperitoneal adipose tissue appears to behave differently from other visceral tissues, resembling subcutaneous adipose tissue. In this way, the different localization sites of adipose tissue in the body seem to perform their functions, including lipolysis and lipogenesis in different ways, as well as responding to external stimuli, such as diet, in different ways. In this regard, other studies corroborate this work, which show that the volume of adipocytes in visceral adipose tissue is greater when compared to those in subcutaneous tissue. Furthermore, adipose tissue depots have different sensitivities to important hormones that regulate adipose tissue metabolism. Subcutaneous adipose tissue appears to be less sensitive to epinephrine and norepinephrine when compared to mesenteric adipose tissue, and may have 50% less lipolysis [35]. Catecholamines are potent activators of lipolysis that act on adrenergic receptors, stimulating hormone-sensitive lipase (HSL) activity and inhibiting LPL [36]. Furthermore, women of childbearing age have greater HSL activity in visceral adipose tissue than in subcutaneous adipose tissue, while in post-menopause, HSL has decreased activity in the former, which may be one of the causes for increased abdominal fat accumulation in women above 50 years old, with consequent redistribution of body composition in this age group [37, 38] .
A study demonstrate that the administration of estradiol in ovariectomized females increased the lipolytic response of adipocytes incubated with isoproterenol, epinephrine, among others, which was accompanied by an increase in cAMP concentration and adenyl cyclase activity [39]. This mechanism is of special importance for the mesenteric adipose tissue because it is densely innervated by the sympathetic nervous system [40], unlike the retroperitoneal which has its own sympathetic innervation, originating mainly from the suprachiasmatic nuclei and the solitary tract [41]. This differentiated sympathetic control may represent an explanation for the differences in responses of these two deposits to similar metabolic stimuli. Furthermore, lipolysis stimulated by the sympathetic nervous system is more effectively inhibited by insulin in adipocytes of mesenteric adipose tissue than in other depots [42]. These observations are in line with the results of this study, which demonstrated different responsiveness of mesenteric adipose tissue when compared to retroperitoneal adipose tissue.
The difference in ER-α expression may suggest a greater estrogen sensitivity in mesenteric adipose tissue than in parametrial adipose tissue, which could explain a greater estrogen-stimulated lipolytic activity in this tissue compared to the other. Indeed, Pedersen and colleagues observed that ER-α was related to the inducing action of estrogen on the expression of alpha 2-adrenergic receptors in adipose tissue [43]. Polymorphisms in the gene for ER-α have been associated with elevated triacylglycerol levels, an increased body mass index and a greater waist circumference, highlighting the role of ER-α in controlling lipid metabolism [44]. Furthermore, mice that do not express ER-α show an increase in adipose tissue mass in the absence of changes in caloric intake, which strongly suggests a role for ER-α in adipose tissue biology [45].
The use of ovariectomized wistar rats, while useful for studying postmenopausal conditions, may not fully replicate human physiology, particularly regarding estrogen effects on adipose tissue and metabolism. In addition, although a high-fat diet was employed to induce obesity, the specific composition of the diet and its potential interaction with other metabolic processes were not thoroughly explored. Moreover, the dosage and administration method of 17β-estradiol may not accurately reflect clinical hormone replacement therapy in humans. The study used a specific dose for a fixed period, which may not account for the variability in hormone levels and effects over longer durations. Finally, the study was limited to 24 weeks of dietary intervention and 14 days of hormone treatment. Longer-term effects and potential changes over extended periods were not assessed.
Thus, the combination of the absence of estrogen, exemplified by ovariectomy, together with a high-fat diet, resulted in a significant increase in the weight and diameter of adipocytes in visceral, parametrial and mesenteric adipose tissues, evidencing the anti-lipogenic and pro-lipolytic influence of estrogen in this context. However, the changes in estrogen receptors after these stimuli were different between the two deposits, suggesting that both expression of the receptors and the nature of the metabolic stimuli may play different roles in these responses. Future studies are needed to clarify the involvement of estrogen receptor isoforms in adipose tissue metabolism. Furthermore, further investigation is critical to understanding the role of divergent responses of visceral fat depots to metabolic stimuli in the pathophysiology of conditions associated with postmenopause, such as diabetes, insulin resistance, and metabolic syndrome.