AUCTORES
Globalize your Research
Research Article | DOI: https://doi.org/10.31579/2688-7517/034
1 Rosa & Co. LLC, San Carlos, CA, USA.
2 Adamis Pharmaceuticals Corp, San Diego Cam, USA.
*Corresponding Author: Ronald B. Moss, Adamis Pharmaceuticals Corporation, San Diego CA. USA.
Citation: Ronald B. Moss, Meghan McCabe Pryor, Rebecca Baillie, Katherine Kudrycki, Christina Friedrich, et al. (2022) Brief Report: Higher Fentanyl Exposures Require Higher Doses of Naloxone for Successful Reversals in a Quantitative Systems Pharmacology Model. J. Addiction Research and Adolescent Behaviour. 5(2); DOI: 10.31579/2688-7517/034
Copyright: © 2022, Ronald B. Moss, This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 21 December 2021 | Accepted: 25 January 2022 | Published: 01 February 2022
Keywords: naloxone; QSP; pharmacology; fentanyl; overdose; higher dose
Background: Previously, we reported on an opioid receptor quantitative systems pharmacology (QSP) model to evaluate naloxone
Methods: In this study, we extended our model to include higher systemic levels of fentanyl (up to 100 ng/ml) and the newly approved 8mg IN naloxone dose (equivalent to 4 mg)
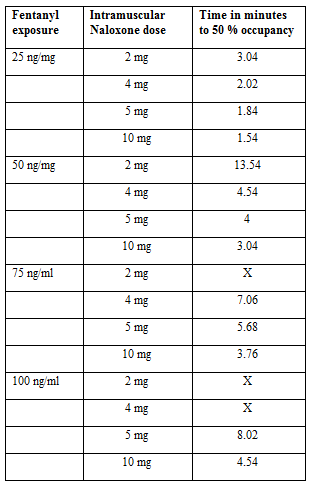
Results: As expected, at the lower peak fentanyl concentrations (25 ng/ml and 50 ng/ml), the simulations predicted that 2 mg, 4 mg, 5 mg, and 10 mg IM doses of naloxone displaced fentanyl and reached below the 50% receptor occupancy within 10 minutes. However, at the concentration of 75 ng/ml, the simulation predicted that the 2 mg dose of naloxone failed to reach below the 50% occupancy within 10 minutes. Interestingly, at the highest peak concentration of fentanyl studied (100 ng/ml), the model predicted that the 4 mg of naloxone IM (equivalent to 8 mg IN) failed to reach below the threshold of 50 % occupancy within 10 minutes or even within 15 minutes (data not shown). In contrast, the model predicted successful reversals when 5 and 10 mg IM doses were utilized.
Conclusion: These results support the notion that acutely administered higher doses of naloxone are needed for rapid and adequate clinical reversal, particularly when higher systemic exposure of the potent synthetic opioids occurs.
Overdoses due to illicit drugs surged in the US during COVID-19, with over 100,000 deaths recently reported by the CDC [1]. Synthetic opioids such as fentanyl continue to be the predominant driver of overdose deaths and are considered to be 50 to 100-fold more potent than morphine [2]. The treatment for effective resuscitation from an opioid overdose is naloxone, a receptor antagonist. The dose of naloxone needed for successful reversal may depend on several factors, including the type of opioid and dose of opioid in the systemic and central nervous system. Previously, we reported on an opioid receptor quantitative systems pharmacology (QSP) model to evaluate naloxone dosing [3]. The previous model predicted naloxone-fentanyl interaction at the mu-opioid receptor and examined three levels of fentanyl exposure and three naloxone doses. All three naloxone doses are FDA approved. At lower fentanyl exposure levels (25 ng/ml and 50 ng/ml), a 2 mg intramuscular (IM) dose of naloxone (equivalent to 4 mg intranasal (IN)) resulted in a time to decrease mu receptor occupancy by fentanyl to 50% within 10 minutes. However, at a higher fentanyl exposure level (75 ng/ml),
2 mg IM dose of the naloxone failed to reduce mu receptor occupancy by fentanyl to 50%.
In contrast, naloxone doses of 5 mg and 10 mg IM reduced mu receptor occupancy by fentanyl to 50% within 10 minutes. However, in some studies, systemic fentanyl levels have demonstrated higher fentanyl exposure post-mortem, with systemic levels reported up to 383 ng/ml [4, 5, 6]. Therefore, it can be speculated that with the current surge in opioid deaths, higher exposure to fentanyl may be playing a role. Concurrent with the recent surges in mortality from synthetic opioids, higher doses of naloxone have now been approved by the US FDA for community use, including an 8 mg IN (Kloxxado, 7) (equivalent to a 4 mg IM dose) and a 5 mg IM dose (Zimhi,8). Thus, we extended our model to include higher systemic levels of fentanyl (up to 100 ng/ml) and the newly approved 8 mg IN naloxone dose (equivalent to 4 mg IM). The results of this model predict that higher doses of IM naloxone are required for
successful overdose reversal when the more potent synthetic opioids such as fentanyl are involved.
A PhysioPD® Research Platform (model) was developed using SimBiology® (The MathWorks, MATLAB® 2018b). The model includes non-linear ordinary differential equations (ODEs) representing plasma and brain pharmacokinetics (PK) for fentanyl and naloxone and mu receptor dynamics. The mu receptor submodel includes standard competitive binding dynamics and accounts for receptor synthesis, internalization, and recycling [3].
Model simulations illustrate the time course of fentanyl concentration in plasma and brain, naloxone concentration in plasma and brain, and fentanyl and naloxone mu receptor occupancy. We assumed peak plasma exposure levels of fentanyl of 25 ng/ml, 50 ng/ml, 75 ng/ml, and 100 ng/ml. Naloxone administration was assumed to occur as a single dose at 5 minutes after peak fentanyl concentration in plasma, which may reasonably reflect response time following initial symptoms.
In addition, we examined time to a reduction of 50% mu receptor occupancy by fentanyl, as this endpoint has been associated with clinical
reversal of opioid toxicity [9]. IN naloxone was assumed to have 50% bioavailability relative to IM [10, 11].
In our model simulations, we examined the time to reach ≤ 50% receptor occupancy by fentanyl due to displacement by naloxone at various systemic levels of fentanyl and multiple doses of IM naloxone (Table 1). As expected, at the lower peak fentanyl concentrations (25 ng/ml and 50 ng/ml), the simulations predicted that 2 mg, 4 mg, 5 mg, and 10 mg IM doses of naloxone displaced fentanyl and reached below the 50% receptor occupancy within 10 minutes (Figure 1a, 1b). However, at the concentration of 75 ng/ml, the simulation predicted that the 2 mg dose of naloxone failed to reach below the 50% occupancy within 10 minutes (Figure 2), as shown previously. Interestingly, at the highest peak concentration of fentanyl studied (100 ng/ml), the model predicted that the 4 mg of naloxone IM (equivalent to 8 mg IN) failed to reach below the threshold of 50 % occupancy within 10 minutes (Figure 3) or even within 15 minutes (data not shown). In contrast, the model predicted successful reversals when 5 and 10 mg IM doses were utilized.
Fentanyl can result in rapid respiratory failure with resulting brain hypoxia and ultimately death. Rapid naloxone systemic treatment, resulting in decreased fentanyl binding to the mu receptors, is critical for a successful reversal and resuscitation.
The idea that higher concentrations of fentanyl exposure may require higher doses of naloxone treatment is supported by data from the field suggesting that multiple administrations of naloxone are required for overdose resuscitations. In one report, the use of multiple administrations of naloxone increased 98% during a 29 week period that included COVID-19 pandemic, compared to the previous period reported by EMS providers [12].
This report, and others [13], support the notion that acutely administered higher doses of naloxone are needed for rapid and adequate clinical reversal, particularly when higher systemic exposure of the potent synthetic opioids occur.
Using an opioid receptor QSP model, we predicted naloxone-fentanyl interaction at the mu-opioid receptor over a range of four naloxone doses. We examined four levels of fentanyl exposure in the plasma found in overdose patients. The high receptor mu occupancy of fentanyl at the doses observed in this model, without naloxone administration, is consistent with reports of the potency of this synthetic opioid and its relation to a recent spike in overdoses and deaths.
The results of model simulations suggest that at higher exposure levels of fentanyl, capable of resulting in respiratory depression and death (75 ng/ml), the currently approved lower doses of naloxone (2 mg IM and 4 mg IN) may be inadequate for a rapid, successful reversal. Most importantly, at the highest exposure levels of fentanyl (100 ng/ml) examined in this simulation, the highest IN dose available (8 mg IN and 4 mg IM) may be inadequate for successful resuscitation. In these simulations, a drop to 50% occupancy at the highest fentanyl exposure was not achieved within 15 minutes with the highest IN dose (8 mg IN 4 mg IM).
The model includes some assumptions but is consistent with other studies suggesting that the mu receptor occupancy by fentanyl increases with an increase in systemic fentanyl exposure and that naloxone receptor occupancy increases as the naloxone dose increases [3].
The model demonstrates that these observations are consistent with a receptor competition mechanism and helps quantify the additional benefit of the higher 5 mg and 10 mg IM doses. These analyses support the idea that higher doses of naloxone (5 mg IM and 10 mg IM), as examined in this model, are superior in many overdose situations when compared to the current naloxone doses (2 mg IM or 4 mg IN, 4 mg IM or 8 mg IN). In this model, higher doses of IM naloxone were superior for a successful resusucitation compared to other available doses and formulations.
These results suggest that with higher fentanyl exposure, higher doses of naloxone may be needed to improve outcomes and decrease morbidity.
Ethics approval and consent to participation:
Not Applicable
Consent to publish:
Yes.
Availability of data and material:
Yes.
Competing interests:
RBM and DJC are employees of Adamis Pharmaceuticals and have equity in the company. MMP, RB, KK, CF, and MR are employees of Rosa & Co and have equity in the company.
Funding
This study was funded by Adamis Pharmaceuticals.
Author contribution
RBM and DJC conceived the hypothesis for the model. MMP, RB, KK, CF and MR developed and ran the model with feedback from RBM and DJC. RBM and DJC wrote the manuscript with comments from MMP, RB, KK, CF, and MR.
Acknowledgement
Not applicable.